Biotech 12 Pack Off to Nice Start..
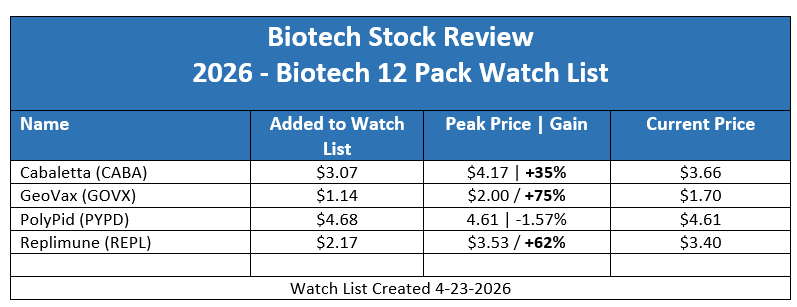
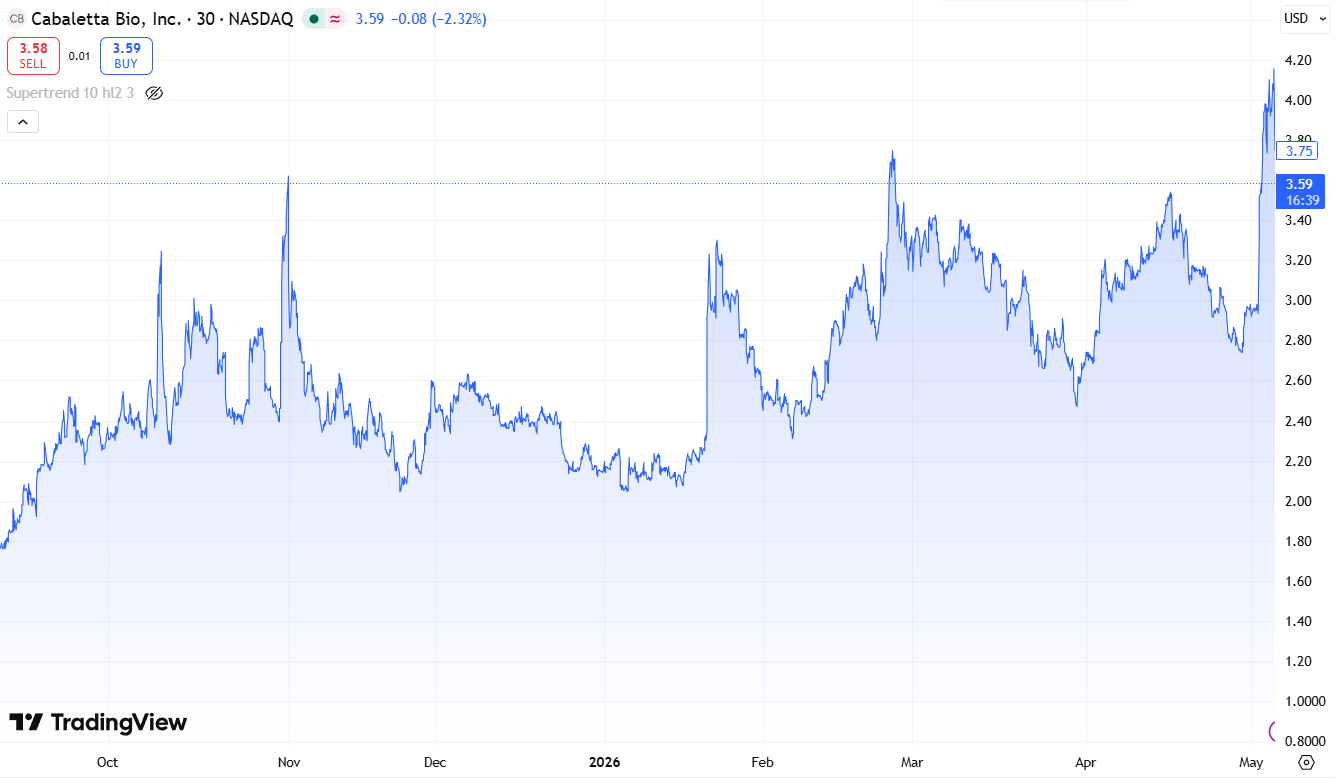
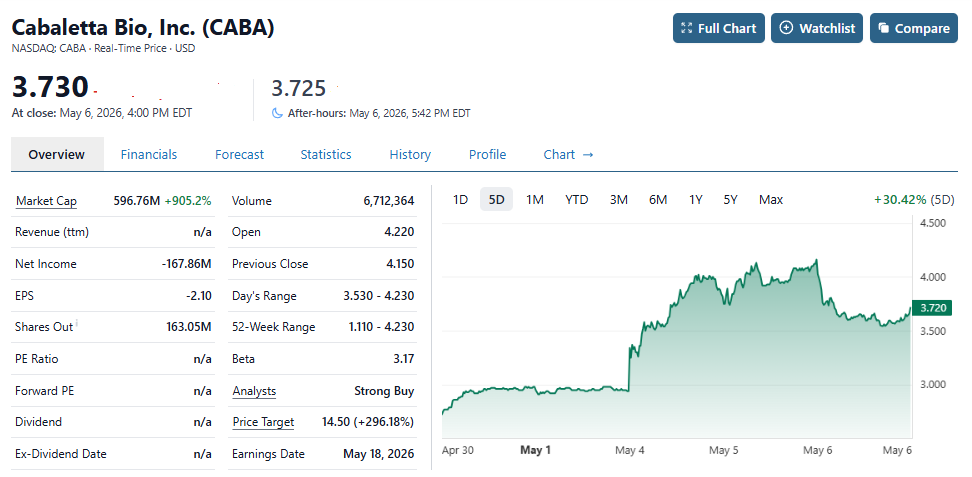
Cabaletta (CABA) Surges 44% on Massive Volume Spike.
A lot of investors are mega-scared of dilution (Cabaletta just sold $155 million worth of shares) and as we’ve tried and tried to explain, that ultimately raising capital is a good thing. No, it’s a great thing. No, it’s the best thing that can happen!
Of course tis all depends on what they spend the funding on..
Worries should instead arise when capital can’t be raised. Cabaletta just proved once again, they have the ability to raise money to advance towards the next stage of their business plan. End of story, with the slight wrinkle of WHO invested.
This round went to Bain Capital Life Sciences, Adage Capital Management, Cormorant Asset Management and other existing investors, multiple new mutual and sovereign wealth funds and Eli Lilly and Company (LLY) . Wait what? Wait who?
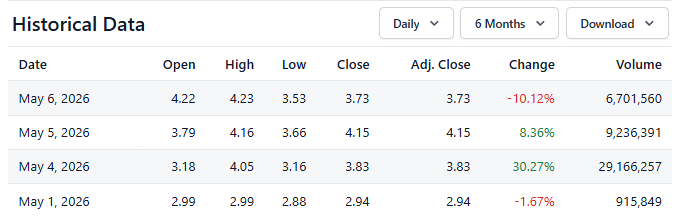
Post offering (@$2.90) it traded as high as $4.23 and closed at $3.73. Burning over $35M in operating cash per quarter, if this was the Indy 500, Cabaletta just gassed up in lap #100, which is enough to get them to lap 150 in a 200 lap race.
Of course the race can end — as it often does for small biotech — on news of a partnership with a major pharma, like (you guessed it) Eli Lilly. Big pharma rarely invests in small clinical‑stage biotechs unless they want optionality: early access to data, a seat at the table, and visibility into manufacturing and clinical progress. Lilly’s participation in this round is the clearest early indicator that they’re tracking Cabaletta’s platform very closely.
Ignore if you want. Cabaletta is as a reminder, highly risky.
We apologize in advance for the ‘tech-talk’ but Cabaletta’s lead asset, rese-cel (CABA‑201), is a CD19-directed CAR‑T for autoimmune diseases — a field Lilly has been aggressively expanding into.
RELATED: Cabaletta Bio Announces Pricing of $150 Million Underwritten Offering
RELATED: Cantor Fitzgerald Reiterates Overweight on Cabaletta Bio, Maintains $30 Price Target
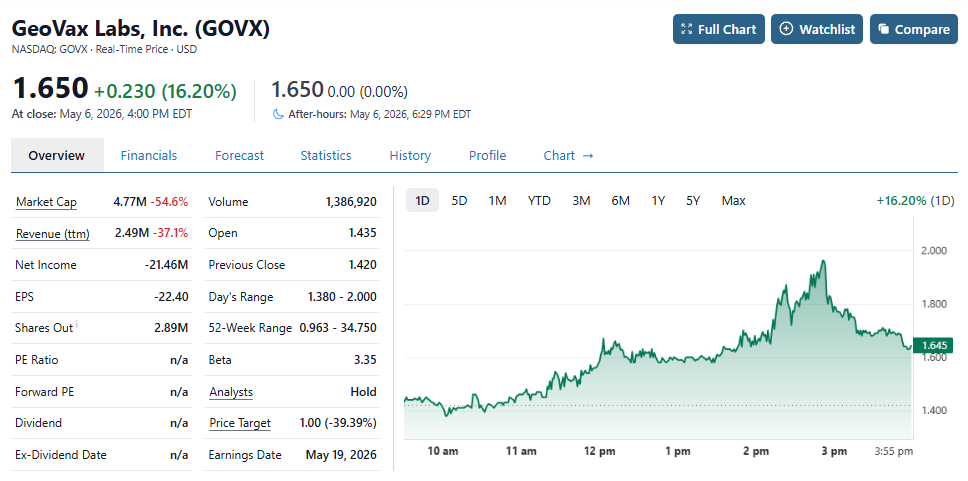
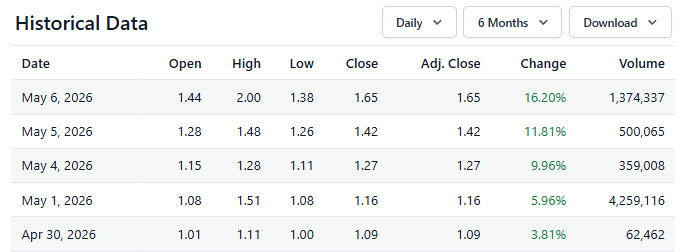
GeoVax (GOVX) Intraday Peak, Gain of 75% Since Adding to Watch List.
Maybe the traders and flippers will stay around past the 7th inning this time. Yes we think they are that advanced into possibly announcing a seminal event, that can come any day. We’re not real sure what ‘seminal’ means, but it just sounds so right. If it does come, we firmly believe it would be related to their MPOX vaccine which is scheduled to go in a head-to-head shootout with Denmark based Bavarian Nordic (BVRNY), near the end of the year.
Bavarian Nordic has generated approximately $2.18–$2.25 billion in cumulative global sales of its MPOX (Jynneos/Imvanex/Imvamune) vaccine since approval, based on all verifiable revenue disclosures from 2022–2024.
Team GeoVax is hungry and after a piece of that huge pie, reminiscent of Samson vs. Goliath. We’re betting on the good old boys from Atlanta, Georgia to kick a little Danish ash. Go Team America!
Related: GeoVax Positions GEO-MVA to Address Supply Constraints in Global Mpox and Smallpox Vaccine Market
Related: GeoVax (GOVX) Surges 51% on Massive Volume Spike and MPOX Related News.
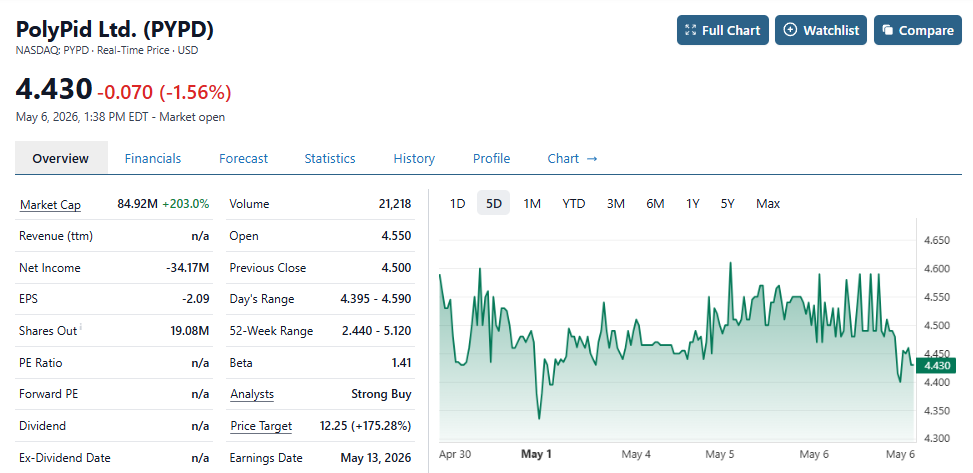
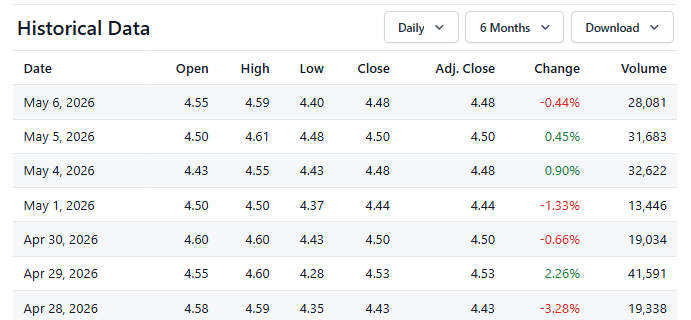
PolyPid to Report First Quarter 2026 Financial Results and Operational Highlights on May 13, 2026
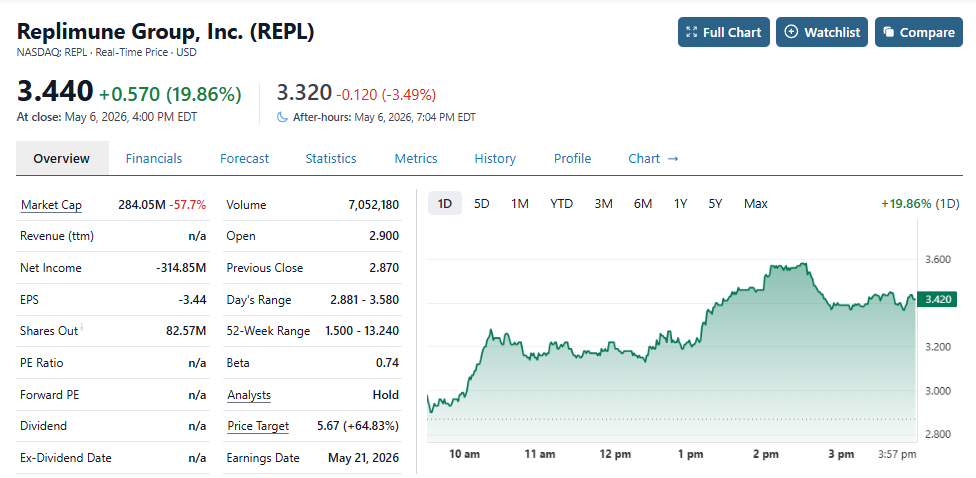
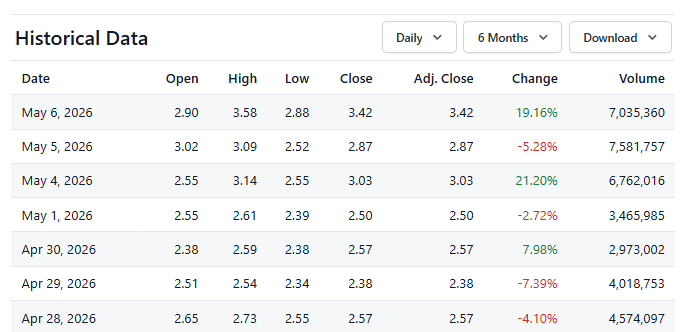
Replimune up 62% From Where we Added to Watch List
RELATED: Replimune Case Fuels Debate Over FDA Review Practices
Past performance is no indication, or inclination, or have anything to do with future performance. Wish it were true, but even we can’t count on our own next idea to perform positively or in a manner we find acceptable lol.
Forward-Looking Statements
CABA: This press release contains “forward-looking statements” of Cabaletta Bio within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including without limitation, express or implied statements regarding: the satisfaction of customary closing conditions related to the offering and sale of securities; the Company’s ability to complete the offering; and use of capital, expenses, future accumulated deficit and other financial results in the future.
Any forward-looking statements in this press release are based on management’s current expectations and beliefs of future events, and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties related to completion of the public offering on the anticipated terms, or at all, include, but are not limited to, market conditions and the satisfaction of customary closing conditions related to the public offering. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause Cabaletta’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in Cabaletta’s most recent annual report on Form 10-K filed on March 23, 2026 and our subsequent quarterly reports on Form 10-Q filed with the SEC, as well as discussions of potential risks, uncertainties, and other important factors in Cabaletta’s other filings with the SEC, including those contained or incorporated by reference in the preliminary prospectus supplement related to the public offering to be filed with the SEC.
GOVX: GeoVax Labs, Inc. is a clinical-stage biotechnology company developing novel vaccines against infectious diseases and therapies for solid tumors. The Company’s lead clinical program is GEO-CM04S1, a next-generation COVID-19 vaccine currently in three Phase 2 clinical trials, being evaluated as (1) a primary vaccine for immunocompromised patients such as those suffering from hematologic cancers and other patient populations for whom the current authorized COVID-19 vaccines are insufficient, (2) a booster vaccine in patients with chronic lymphocytic leukemia (CLL) and (3) a more robust, durable COVID-19 booster among healthy patients who previously received mRNA vaccines. In oncology, the lead clinical program is evaluating a novel oncolytic solid tumor gene-directed therapy, Gedeptin®, having recently completed a multicenter Phase 1/2 clinical trial for advanced head and neck cancers. GeoVax is also developing a vaccine targeting Mpox and smallpox and, based on recent EMA regulatory guidance, anticipates progressing directly to a Phase 3 clinical evaluation, omitting Phase 1 and Phase 2 trials. GeoVax has a strong IP portfolio in support of its technologies and product candidates, holding worldwide rights for its technologies and products. For more information about the our clinical trials and other updates, visit our website: www.geovax.com.
Forward-Looking Statements
This release contains forward-looking statements regarding GeoVax’s business plans. The words “believe,” “look forward to,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax is able to obtain acceptable results from ongoing or future clinical trials of its investigational products, GeoVax’s immuno-oncology products and preventative vaccines can provoke the desired responses, and those products or vaccines can be used effectively, GeoVax’s viral vector technology adequately amplifies immune responses to cancer antigens, GeoVax can develop and manufacture its immuno-oncology products and preventative vaccines with the desired characteristics in a timely manner, GeoVax’s immuno-oncology products and preventative vaccines will be safe for human use, GeoVax’s vaccines will effectively prevent targeted infections in humans, GeoVax’s immuno-oncology products and preventative vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete development, there is development of competitive products that may be more effective or easier to use than GeoVax’s products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control.
Further information on our risk factors is contained in our periodic reports on Form 10-Q and Form 10-K that we have filed and will file with the SEC. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as may be required by law. GeoVax is a client of Institutional Analyst (and not Katogen), the publisher of the Biotech Stock Review and has been retained with a combination of cash and equity for ongoing progress, news coverge and research reporting for five thousand dollars per month.