In our view, a research report is designed to gather information on publicly traded companies from press releases, management presentations (including power points), SEC filings, management guidance, industry outlooks, product or service descriptions and stock charts – to name a few and then present all of the information in one neat package.
The point is to gather all the information that may require an investor visiting a dozen different financial sites to gather on their own, and put it one nice, neat package – so they don’t have to bounce from site to site.
Of course in some reports investors aren’t looking for information so they can make a decision on their own. They are looking to be told what to do. That’s where price targets and buy or sell edicts are presented. And there is nothing wrong with that.

In any event we came across an AI Analyst so to speak called Katogen Engine lead by Doug Drysdale*, former CEO of Cybin now rebranded to Helus (HELP).
So we took it for a spin. In the query box below we simple entered “Can I please get a summary report designed to give an investor an overview of GeoVax symbol GOVX.”
And in about 60 seconds – NICE JOB!
Katogen Engine is now a perma-tool we’ll add to our box of analytical toys. We won’t rely on it solely of course, but we’ll run it every time we find a new idea (a dozen every week), read what the report overview says, then collect our thoughts, and then decide whether to proceed with further more time consuming research.
It doesn’t (yet – we’ve only used it once) function as a bird dog for us. We’ll continue to rely on investment bankers, hedge fund managers and certain ‘smart-people’ we know for that.
You can get three free reports a month, or unlimited for just $49 a month. A bargain considering all the time it will save. We could easily select or eliminate 10 companies a day for further consideration using it.
Try it yourself, on your favorite Biotech. We went to the page below and typed in our query “Can I please get a summary report designed to give an investor an overview of GeoVax symbol GOVX” and then selected one of 7 reports you can choose from.
CLICK HER TO RUN YOUR OWN REPORT
And voilà 60 seconds later..
CLICK HERE TO VIEW GEOVAX REPORT
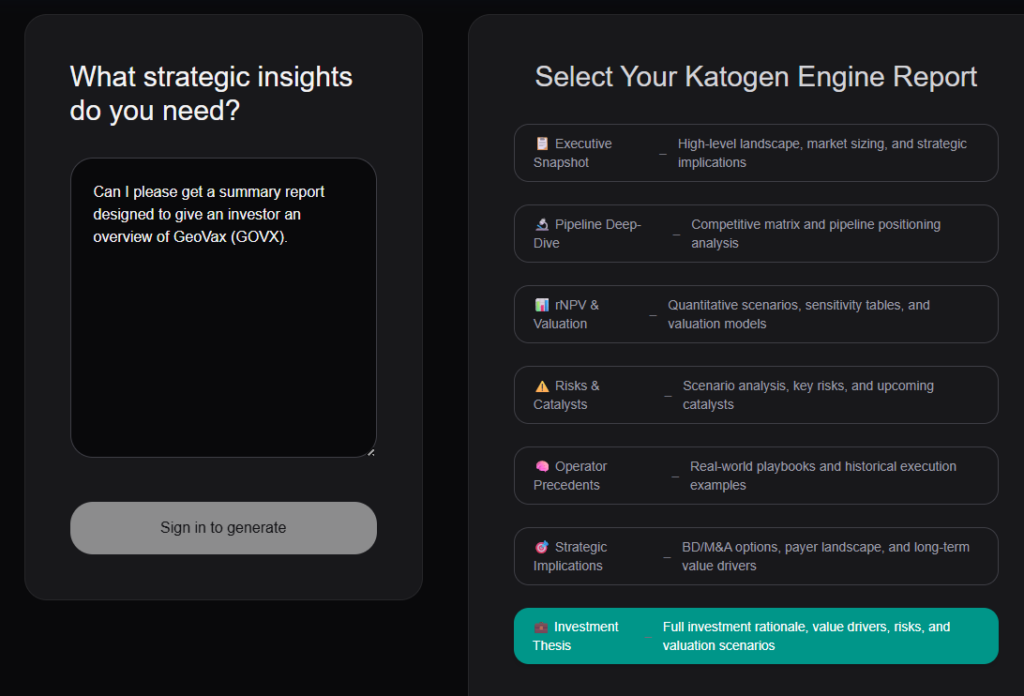
The Katogen Engine also just got a MASSIVE upgrade. Seven strategic reports are now available. Professional-caliber analysis in 60 seconds without the six-figure invoice.
Here’s what you can get:
1) Executive Snapshot
2) Pipeline Deep-Dive
3) rNPV & Valuation
4) Risks & Catalysts
5) Operator Precedents
6) Strategic Implications
7) Investment Thesis
The Engine is a rapid strategic intelligence tool built for biopharma. Query an asset, a deal structure, a reimbursement scenario, a competitive position. Get a structured, rigorous report in about a minute. This runs alongside Katogen’s bespoke advisory practice, which is grounded in 35 years of biopharma operator experience, and two decades in the CEO seat.
The advisory work is deliberate, deep, and built for complex situations that require judgment, not just information. The Engine is built for speed and access. Both exist because biopharma decisions rarely wait.
Free tier: 3 reports per month. Pro tier: $49/month for unlimited access.
*Doug Drysdale. Over nearly two decades as a CEO, Doug built and turned around four companies – two public and two private – leading 17 acquisitions across three continents and raising over $4.5 billion in capital. As CEO of Cybin, he took the company from molecule inception to Phase 3 trials in just three years, advancing breakthrough therapies in mental health. Previously, as Founding CEO of Alvogen, he scaled the company to $500 million in revenue across 35 countries in under six years. At Pernix Therapeutics, he led a successful turnaround, growing enterprise value from $80 million to $800 million, and earlier in his career, he drove global M&A at Actavis, leading 15 acquisitions and raising $3 billion.
Disclaimer. AI can and will make mistakes, just like us.
About GeoVax
GeoVax Labs, Inc. is a clinical-stage biotechnology company developing novel vaccines against infectious diseases and therapies for solid tumors. The Company’s lead clinical program is GEO-CM04S1, a next-generation COVID-19 vaccine currently in three Phase 2 clinical trials, being evaluated as (1) a primary vaccine for immunocompromised patients such as those suffering from hematologic cancers and other patient populations for whom the current authorized COVID-19 vaccines are insufficient, (2) a booster vaccine in patients with chronic lymphocytic leukemia (CLL) and (3) a more robust, durable COVID-19 booster among healthy patients who previously received mRNA vaccines. In oncology, the lead clinical program is evaluating a novel oncolytic solid tumor gene-directed therapy, Gedeptin®, having recently completed a multicenter Phase 1/2 clinical trial for advanced head and neck cancers. GeoVax is also developing a vaccine targeting Mpox and smallpox and, based on recent EMA regulatory guidance, anticipates progressing directly to a Phase 3 clinical evaluation, omitting Phase 1 and Phase 2 trials. GeoVax has a strong IP portfolio in support of its technologies and product candidates, holding worldwide rights for its technologies and products. For more information about the our clinical trials and other updates, visit our website: www.geovax.com.
Forward-Looking Statements
This release contains forward-looking statements regarding GeoVax’s business plans. The words “believe,” “look forward to,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax is able to obtain acceptable results from ongoing or future clinical trials of its investigational products, GeoVax’s immuno-oncology products and preventative vaccines can provoke the desired responses, and those products or vaccines can be used effectively, GeoVax’s viral vector technology adequately amplifies immune responses to cancer antigens, GeoVax can develop and manufacture its immuno-oncology products and preventative vaccines with the desired characteristics in a timely manner, GeoVax’s immuno-oncology products and preventative vaccines will be safe for human use, GeoVax’s vaccines will effectively prevent targeted infections in humans, GeoVax’s immuno-oncology products and preventative vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete development, there is development of competitive products that may be more effective or easier to use than GeoVax’s products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control.
Further information on our risk factors is contained in our periodic reports on Form 10-Q and Form 10-K that we have filed and will file with the SEC. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as may be required by law. GeoVax is a client of Institutional Analyst (and not Katogen), the publisher of the Biotech Stock Review and has been retained with a combination of cash and equity for ongoing progress, news coverge and research reporting for five thousand dollars per month.
Company Contact:
info@geovax.com
678-384-7220
Media Contact:
Jessica Starman
media@geovax.com