Psycheceutical Bioscience, Inc. Is Developing a Topical Application Of Ketamine Targeted to Relieve PTSD Symptoms Within Minutes and Negate Side Effects
FT. LAUDERDALE, FL / ACCESSWIRE / April 18, 2023 / Psycheceutical Bioscience, Inc (OTC PINK:BWVI) is a biotechnology company developing a new and unique mode of administration that has the potential to completely revolutionize the psychedelic mental health space. With a staggering 52.9 million adults living with mental illness in the U.S. alone and current treatments for mental health still using 1980s technology, millions of people have been left with no viable options to treat these debilitating diseases.
While selective serotonin reuptake inhibitors (SSRIs) have been the mainstay for mental health treatment since the 1980s, they’re not actually designed for long-term use and can have severe withdrawal symptoms. And in fact, new research has shown no evidence that low serotonin levels cause depression. Meanwhile, 1 in 6 American adults has reported taking a psychiatric drug, with the most widely prescribed form of antidepressant being an SSRI.
Psychedelics To The Rescue
However, there is a growing interest and demand for the use of psychedelics for mental health treatment. The therapeutic benefits of psychedelics – especially the Schedule 3 compound ketamine – for treating mental health conditions like PTSD, anxiety, and depression have shown promising results in preclinical and clinical trials.
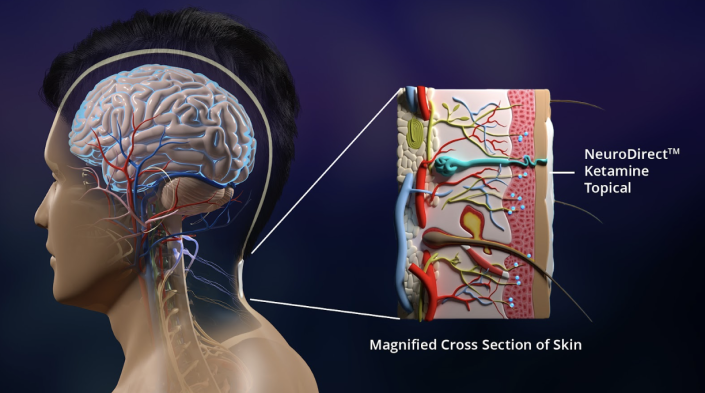
To meet the desperate need for safe psychedelic medicines, Psycheceutical is developing a topical cream administration of ketamine which is designed to allow the drug to directly target the brain and bypass the GI tract and liver. This process is believed to eliminate the hallucinogenic effects of psychedelic drugs and significantly reduce the toxicity and adverse effects of these compounds.
Currently, the ketamine treatment market is limited to intravenous (IV) or oral delivery. While effective, the cost of treatments like ketamine infusions can be between $400 and $2000 per infusion. Not only are these treatments expensive, but they also normally require a clinical setting for administration and monitoring. Side effects such as nausea, headaches, dizziness, disorientation and confusion are common with oral and IV ketamine routes of administration. Some patients also report experiencing unwanted “psychedelic” effects when receiving treatment, leaving a huge opportunity for more patient-friendly treatment options.
Solution In A Topical Cream
Psycheceutical’s patented NeuroDirect™ ketamine topical cream is being developed to address the unmet need for rapid relief of PTSD symptoms. The cream is designed to act within minutes, and be administered at home instead of a clinical setting, which would greatly lower the cost of care for both insurance companies and patients and increase access to these life-saving treatments for anyone suffering from mental health disorders.
Psychedelic pioneer and Founder and Executive Director of the Multidisciplinary Association for Psychedelic Studies (MAPS), Rick Doblin, recently visited one of Psycheceutical’s labs to meet their team and learn more about their novel ketamine cream. Afterwards he shared, “I’ve been working in psychedelics for more than 40 years, and it’s rare for me to come across something unique like Psycheceutical’s NeuroDirect topical cream that’s being developed to deliver a psychedelic compound without a psychedelic experience or negative side effects. I can imagine there are a lot of implications.” He also expressed an interest in seeing if the company’s patented technology could be applied to other drugs like MDMA. “Sometimes the biggest breakthroughs are right under our noses. I look forward to exploring it.”
Not only is the NeuroDirect cream an innovative idea, but it’s already seeing positive results in observational clinical data. A peer-reviewed study published by Drug Development & Delivery shared that more than 80% of 100 PTSD patients that tried NeuroDirect topical ketamine cream found it effective at treating their symptoms, with no patients experiencing psychogenic effects like hallucinations. In fact, patients felt more focused and felt that their thought processes were clearer, with one patient even describing the treatment as “[A] truth serum! I became aware of thoughts and emotions I had suppressed which needed to come out.”
Relief Anywhere, Anytime
One major issue that Psycheceutical is working to solve is “the treatment gap,” a critical shortage of mental health practitioners to meet the overwhelming demand for mental health services. A second issue is the lack of trained psychedelic therapists for those seeking psychedelic treatment for their mental health.
Not only is a psychedelic “trip” not desired by many who would otherwise seek these innovative treatments, but it’s estimated that more than 8,000 therapists are needed in the U.S. the next decade alone to specifically help administer psychedelic compounds. Psycheceutical believes its technology will make it possible for patients to receive medication in a manner that reduces or completely mitigates the systemic side effects of psychedelic compounds, which could potentially open up a new world for treating mental health conditions.
With clinical trials for Psycheceutical’s NeuroDirect topical ketamine set to begin in Australia soon, this new way of administering psychedelic medicine holds very exciting promise for mental health treatment. While it’s clear that ketamine and psychedelic treatments for mental health illnesses are here to stay, Psycheceutical is working to develop its ketamine treatments to provide a safer, more accessible and more affordable option for people struggling with PTSD.
Visit www.psycheceutical.com for more information on the company.
Featured photo by Robina Weermeijer on Unsplash
Contact:
Kaia Roman, VP Strategy and Communications
kaia.roman@psycheceutical.com
SOURCE: Psycheceutical Bioscience, Inc.#BWVI, $BWVI












