We’re up 126% Since December, and That Ain’t Bad.
Note: This isn’t the end, it’s just the beginning.
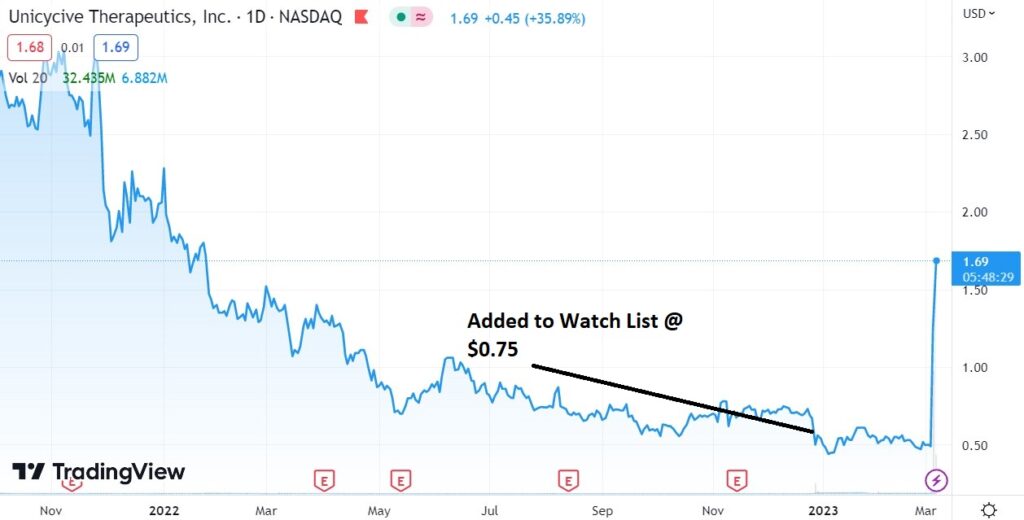
Adding Unicycive (UNCY) $0.75 to the Watch List.
Unicycive Announces up to $130 Million Financing to Commercialize and Launch Investigational New Drug Renazorb
March 06, 2023 07:00 ET | Source: Unicycive Therapeutics, Inc.
$30 million financing upfront with up to an additional $100 million tied to satisfaction of milestones
Led by Vivo Capital with participation from RA Capital, BVF Partners, Logos Capital, and supported by existing investors Nantahala Capital Partners and Rosalind Advisors Inc.
Vivo Capital’s Gaurav Aggarwal, MD to join the Board
Company to implement mandatory dividend policy upon achievement of profitability
LOS ALTOS, Calif., March 06, 2023 (GLOBE NEWSWIRE) — Unicycive Therapeutics, Inc. (Nasdaq: UNCY) (the “Company” or “Unicycive”), a clinical-stage biotechnology company developing therapies for patients with kidney disease, today announced that the Company has signed a securities purchase agreement with certain healthcare-focused institutional investors that will provide up to $130 million in gross proceeds to Unicycive through a private placement that includes initial upfront funding of $30 million.
The funding is being led by Vivo Capital with participation from RA Capital, BVF Partners, Logos Capital, and is supported by existing investors Nantahala Capital Partners and Rosalind Advisors Inc. In conjunction with the financing, Gaurav Aggarwal, M.D., Managing Director of Vivo Capital, will join the Unicycive Board of Directors.
“We are delighted to have the financial backing of both existing and new investors from these high profile, healthcare-focused funds as it provides strong validation of the best-in-class potential for Renazorb® and provides funding for its potential commercial launch, if approved. We remain on track to file a New Drug Application (NDA) for Renazorb marketing approval with the U.S. Food and Drug Administration (FDA) in mid-year,” said Shalabh Gupta, M.D., Chief Executive Officer of Unicycive.
Commenting on their commitment to Unicycive, Dr. Aggarwal said, “We believe that dialysis patients are not well served with the phosphate lowering agents currently on the market, either due to high pill burden, suboptimal tolerability profiles or both. We are pleased to support Unicycive as it nears NDA submission and prepares to launch Renazorb, a phosphate binder that potently controls phosphate with only three small tablets per day. In addition, we have structured an investment that enables capital availability for the Company at increasing share prices upon achievement of milestones with a unique feature that mandates dividends to all shareholders as the company generates profitability.”
In December 2022, Unicycive announced that the primary endpoint was met in the Company’s pivotal bioequivalence (BE) study comparing Renazorb to Fosrenol®. Based on the topline results, pharmacodynamic (PD) BE of Renazorb to Fosrenol was established and met FDA’s regulatory criteria for PD BE in the healthy volunteer study. Unicycive previously received confirmatory guidance from the FDA that this single BE study in healthy volunteers would satisfy all clinical regulatory requirements and that no other clinical studies would be required for an NDA filing through the 505(b)(2) pathway.
“If approved, Renazorb may dramatically reduce the pill burden that patients endure with currently available medications. The global market opportunity for treating hyperphosphatemia is projected to be in excess of $2.5 billion in 2023, with the United States accounting for more than $1 billion of that total. Despite the availability of several FDA-cleared medications, 75 percent of U.S. dialysis patients fail to achieve the target phosphorus levels recommended by published medical guidelines. The timing of Renazorb’s expected launch coincides favorably with the pending expansion of Medicare patient access to phosphate binders in 2025 when these products are added to the ESRD PPS through the Center for Medicare and Medicaid’s Transitional Drug Add-On Payment Adjustment (“TDAPA”) program,” added Dr. Gupta.
About the Private Placement
Pursuant to the securities purchase agreement, the Company will issue to purchasers (i) $30 million in shares of the Company’s Series A Convertible Preferred Stock and (ii) three tranches of warrants that are exercisable for convertible preferred stock as follows:
- The Tranche A warrants for an aggregate exercise price of approximately $25 million are exercisable until 21 days following the Company’s announcement of receipt of FDA approval for Renazorb;
- The Tranche B warrants for an aggregate exercise price of approximately $25 million are exercisable until 21 days following the Company’s announcement of receipt of TDAPA approval for Renazorb; and
- The Tranche C warrants for an aggregate exercise price of approximately $50 million are exercisable until 21 days following public disclosure of four quarters of commercial sales of Renazorb following receipt of TDAPA approval.
Shares of Series A Convertible Preferred Stock will be issued at a price of $1,000.00 per share. All of the shares of preferred stock, the warrants and the securities underlying such shares of preferred stock and warrants, are being offered by Unicycive.
Proceeds from the offering will be used to support the Company’s NDA submission with the FDA for approval of Renazorb for the treatment of hyperphosphatemia in the U.S. and, if approved, for the commercial launch of Renazorb in the U.S.
In addition, the Company shall modify its dividend policy to state that the Company intends to pay dividends to all stockholders, including holders of Series A Preferred Stock on an as-if-converted-to-common-stock basis, on a quarterly basis in an amount of which the aggregate of all quarterly dividends shall equal at least seventy-five percent (75%) of its annual net cash flow from operations following the approval of Renazorb by the FDA if obtained, and the commencement of commercial sales.
EF Hutton, division of Benchmark Investments, LLC acted as the sole placement agent for the offering. Maxim Group LLC acted as financial advisor to the Company.
The securities to be issued in connection with the offering described above are being offered in a private placement under Section 4(a)(2) of the Securities Act of 1933, as amended (the “Act”), and Regulation D promulgated thereunder and have not been registered under the Act or applicable state securities laws. Accordingly, such securities may not be offered or sold in the United States except pursuant to an effective registration statement or an applicable exemption from the registration requirements of the Act and such applicable state securities laws.
This press release shall not constitute an offer to sell or a solicitation of an offer to buy any of the securities described herein, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such state or other jurisdiction.
For further information, please see the Company’s current report on Form 8-K to be filed with the Securities and Exchange Commission.
About Unicycive Therapeutics
Unicycive Therapeutics is a biotechnology company developing novel treatments for kidney diseases. Unicycive’s lead drug, Renazorb, is a novel investigational phosphate-binding agent being developed for the treatment of hyperphosphatemia in chronic kidney disease patients on dialysis. UNI-494 is a patent-protected new chemical entity in late preclinical development for the treatment of acute kidney injury. For more information, please visit www.unicycive.com.
Forward-looking statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified using words such as “anticipate,” “believe,” “forecast,” “estimated” and “intend” or other similar terms or expressions that concern Unicycive’s expectations, strategy, plans or intentions. These forward-looking statements are based on Unicycive’s current expectations and actual results could differ materially. There are several factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, clinical trials involve a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results; our clinical trials may be suspended or discontinued due to unexpected side effects or other safety risks that could preclude approval of our product candidates; risks related to business interruptions, including the outbreak of COVID-19 coronavirus, which could seriously harm our financial condition and increase our costs and expenses; dependence on key personnel; substantial competition; uncertainties of patent protection and litigation; dependence upon third parties; and risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties related to market conditions and other factors described more fully in the section entitled ‘Risk Factors’ in Unicycive’s Annual Report on Form 10-K for the year ended December 31, 2021, and other periodic reports filed with the Securities and Exchange Commission. Any forward-looking statements contained in this press release speak only as of the date hereof, and Unicycive specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events or otherwise.
Investor Contact:
ir@unicycive.com
(650) 900-5470
Anne Marie Fields
Stern Investor Relations
annemarie.fields@sternir.com
212-362-1200
SOURCE: Unicycive Therapeutics, Inc.












