Stock Levitates After Series of Positive Press Releases on Zofrin.
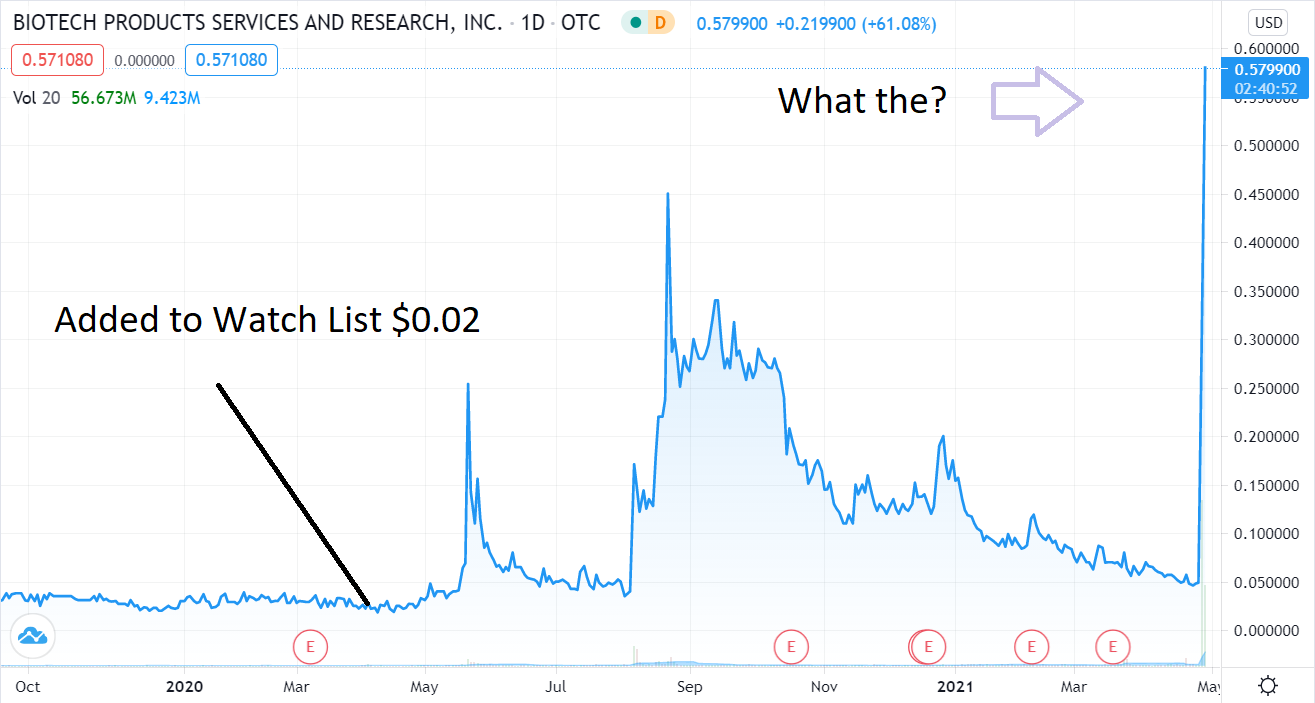
Added to the Watch List in April a little more than a year ago after we met management (Albert Mitrani) in Miami Beach back when travel to Miami was like visiting a ghost town. We met at his home to play it safe. At the time trading at $0.02 – it had a market value of near $12 million with 550 million shares outstanding. Which seemed rich, plus they were on the Pink Sheets at the time..
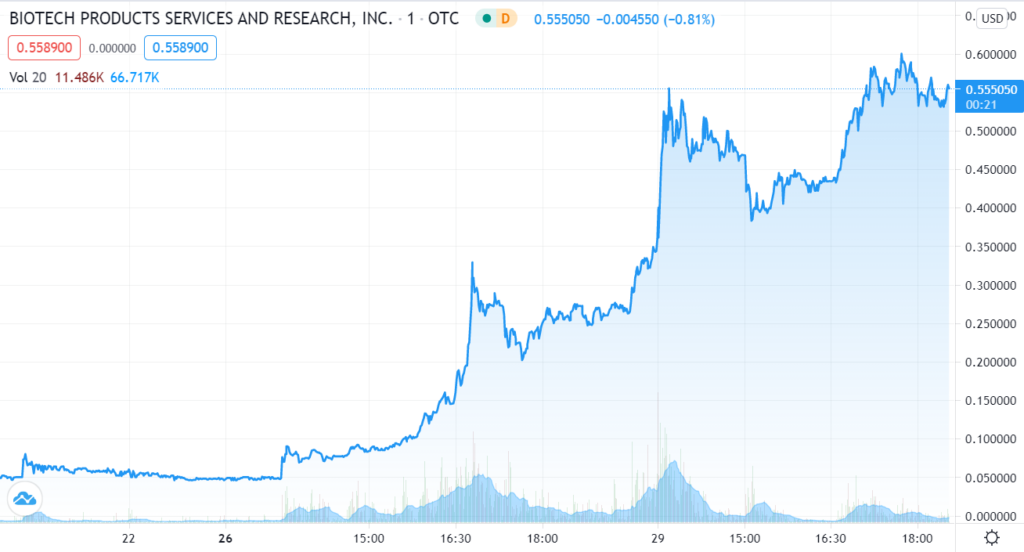
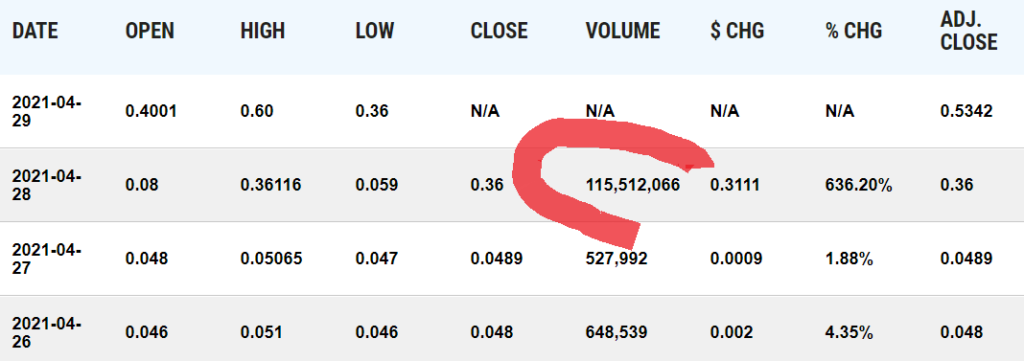
Today, with one BILLION shares outstanding, it hit a market value of $500 million. Wait, what? That’s a 4,066% gain in one year. We’ll just bet some lucky fella made bank because they weren’t around to sell

The first mention of Zofrin in a serious light, came in March of 2020 when ‘Frontiers in Medicine’ published a case report on their exciting results of Zofin treatment for respiratory failure induced by COVID-19 infection.
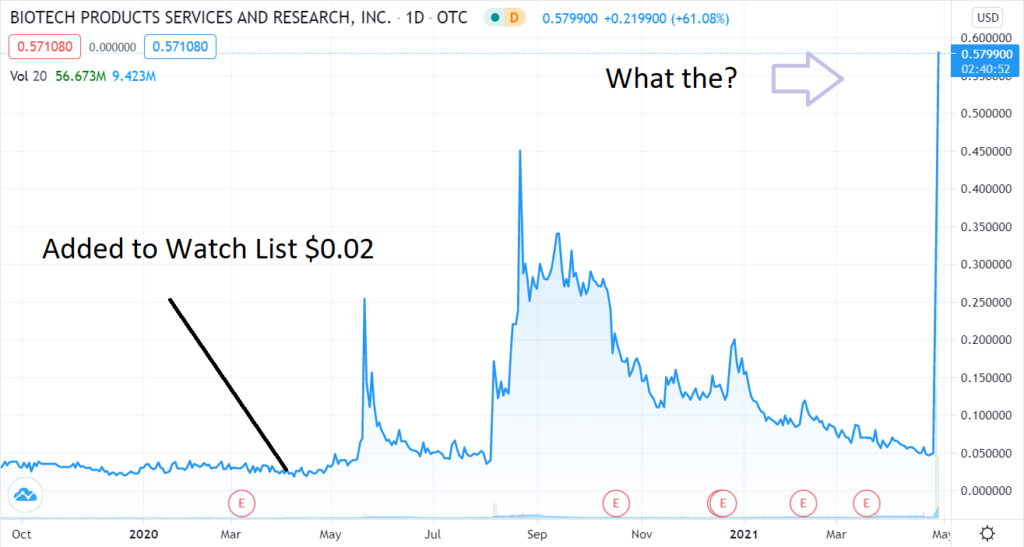
Adding Organicell (BPSR) $0.02 to the Watch List.
Here are the recent headline-makers, which can account for some of the move. The moral of the story is, you should have some Stem Cells in your portfolio, as well as undergo a stem cell treatment like we did – from Pacific Stem a subsidiary of Dalrada (DFCO) a West Coast-based med-related incubator.
The stuff really works. We spent time with management who was once again in Florida last week when they came to visit us and to treat patients. The results they are getting are simply mind-boggling and we feel 20 (maybe 30) years younger.
HEADLINES (older first):
4/22: Organicell Completes Enrollment for its Expanded Access Trial to Treat COVID-19 Patients with Zofin™
And then yesterday:
The initial ten trial participants were hospitalized patients and were treated at Narayana Hrudayalaya Hospital in Bangalore, Lisa Hospital in Kozhikode, Kerala and Prime Indian Hospital in Chennai, and all patients have recovered from their symptoms and have since been discharged from the hospital. Based on the initial results of this trial, the trial will be expanded to an additional sixty-five patients with moderate to severe COVID-19, who will be treated at these hospitals over the next several weeks, with treatments expected to be completed by the end of June 2021.
If the results of the expanded trial are similarly positive, Organicell and CWI India, intend to file with the ICMR (Indian Council for Medical Research) for Emergency Use Approval to use Zofin™ in India as a therapeutic for treating COVID-19.
The second moral of the story is when we say “look what we found or we found something cool” pay attention. Get it, got it? We’re gone.
The big picture we see with stem cells is because of all of the skepticism – and not despite all of the skepticism, there are fantastic opportunities for stem cell providers and fantastic opportunities stem cell investors – because big pharma is scared to tread here.
It’s really that simple. We’ll relaunch our Stem Cell stock newsletter next week. We have over 25 companies in our database. We first launched it in 2015 and even sponsored a Stem Cell conference, but we were way too early so we moth-balled it and cried (chuckled actually) over a few Martinis. It was an expensive endeavor.
But now we’re back. And we’re are going to be the King of Stem Cell stocks!
Just don’t call it a comeback, we been here for years. We’re gonna knock you out.