Research Report Archive
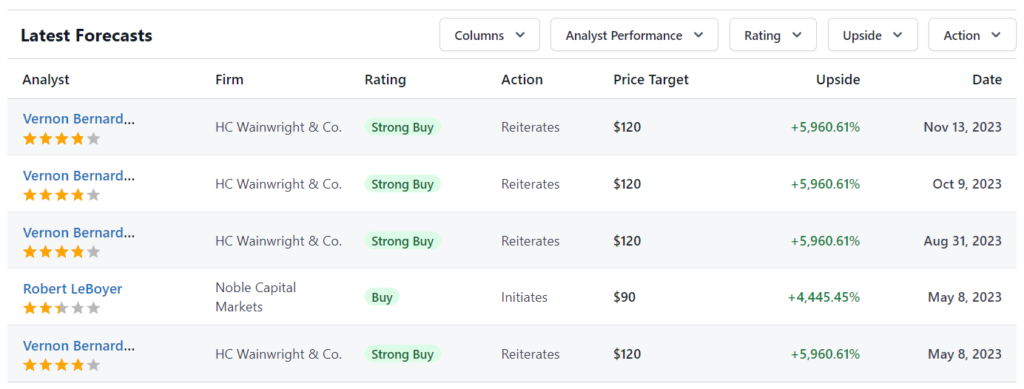
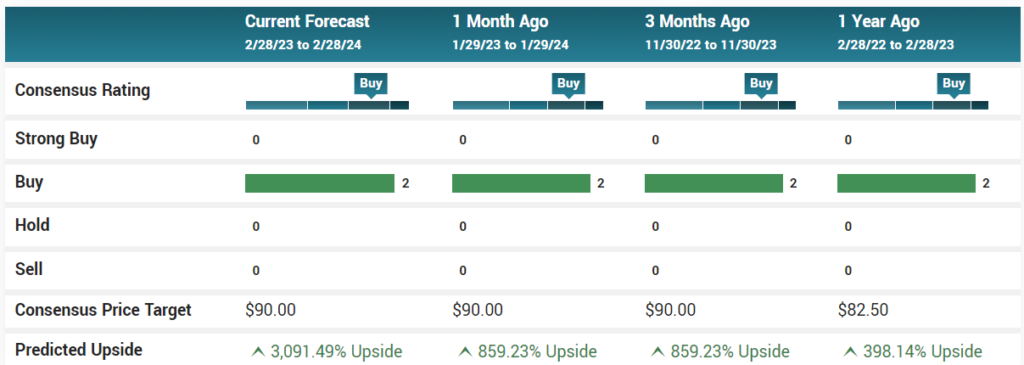
Reports from Robert LeBoyer at Noble Capital. Jeffrey Kraws and Karen Goldfarb at Crystal Research. Jason Kolbert at Dawson James Securities. Vernon Bernardino at H.C. Wainwright. Various price targets.


May 2024
1Q24 Reported With Clinical Milestones Later In FY2024

Killer, 44-Page Report on GeoVax (GOVX). March, 2024

December 22nd, 2023 Biotech Stock Review.
Adding GeoVax (GOVX) to 2024 Biotech Watch List.
WALL STREET RESEARCH
(*Crystal Research’s reports are the most comprehensive)

CRYSTAL RESEARCH ASSOCIATES WEBSITE
August 19th, 2023 Noble Capital. $6 Price Target.

NOBLE CAPITAL MARKETS WEBSITE:
August 11th, 2023 Crystal Research.

August 10th, 2023 Dawson James, $4.00 price target.

DAWSON JAMES SECURITIES WEBSITE
May 8th, 2023 HC Wainwright & Co. $8 Price Target.

H.C. WAINWRIGHT & CO. WEBSITE
May 8th, 2023 Noble Capital.$6 Price Target.

March 3rd, 2023 Dawson James. $4 Price Target

October 26th, 2022 HC Wainwright & Co. $8 Price Target.


Most recent S1-Filing, a mandatory read for investors conducting due diligence.
GeoVax Labs, Inc. is followed by the analysts listed above. Please note that any opinions, estimates or forecasts regarding GeoVax Labs, Inc.’s performance made by these analysts are theirs alone and do not represent opinions, forecasts or predictions of GeoVax Labs, Inc. or its management. GeoVax Labs, Inc. does not by its reference below or distribution imply its endorsement of or concurrence with such information, conclusions or recommendations.
Disclaimer, neither the Company nor Institutional Analyst Inc. (IA) approve or disapprove of the findings or conclusions of the above reports, which are solely that of the respective author|analysts(s). Please see respective disclaimers of each report for disclosure and relationship details. Summary charts updated monthly. IA will make best effort to update and post new reports, but is under no obligation top do so.
Forward-Looking Statements
This post contains forward-looking statements regarding GeoVax’s business plans. The words “believe,” “look forward to,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax is able to obtain acceptable results from ongoing or future clinical trials of its investigational products, GeoVax’s immuno-oncology products and preventative vaccines can provoke the desired responses, and those products or vaccines can be used effectively, GeoVax’s viral vector technology adequately amplifies immune responses to cancer antigens, GeoVax can develop and manufacture its immuno-oncology products and preventative vaccines with the desired characteristics in a timely manner, GeoVax’s immuno-oncology products and preventative vaccines will be safe for human use, GeoVax’s vaccines will effectively prevent targeted infections in humans, GeoVax’s immuno-oncology products and preventative vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete development, there is development of competitive products that may be more effective or easier to use than GeoVax’s products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control.
Further information on our risk factors is contained in our periodic reports on Form 10-Q and Form 10-K that we have filed and will file with the SEC. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as may be required by law. GOVX is a client of Institutional Analyst which receives a monthly retainer of five-thousand dollars for ongoing progress reporting and news coverage.














