Full Report Monday!
And we’re not letting this spoil our Christmas..

But yes, it sucks, and yes, we’ve been here before. And no, we were not originally intending on spending our entire pre-holiday weekend, writing up GeoVax. But it is what it is. As they say “No Man Left Behind.”
Before today’s ‘event’ we were already planning on releasing a report Monday, but we have been deluged by some of our bestest best, and closest friends – who unlike us are panicking. Our blanket statement is “we love the company more today, than we did last week.” Full stop (love that saying).
What we mean by “we’ve been here before” is being in a siutation where a grenade goes off, with something we love.
What we’ve learned to do, is pretend this is the very first day we’ve ever heard of the stock (this goes for stocks up 100 or 200% in a day too) and look at it with a fresh eye. Then ask ourselves with all of the current information we have in hand – do we sit pat, watch and stay, buy more or sell?
Then we act accordingly with our fresh and brand new opinion.
Short story is they had three huge back to back major news annoucements in the last week, and then one tiny raise for $3.2 million. As we’ve said many, many times before, GeoVax and ALL Biotech companies will continue to raise money right up until judgement day. It’s something we’ve grown to live with. It is gas to fuel the race car.
We could say everything is going as planned, but in reality (aside from the stock price) everything is going BETTER than planned. Read the news and see for yourself.
See you Monday.
GeoVax to Raise Approximately $3.2 Million of Gross Proceeds in Public Offering
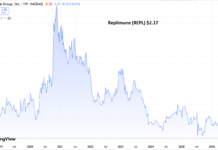
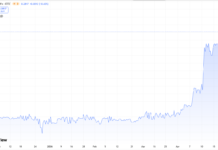
With the additional shares outstanding, we would expect some of the loftier price targets to come down a bit..but we’re still talking about once in a life time potential returns.
See TipRanks below, with Roth, their underwriter with a $14 price target. This of course assumes one of their three shots on goal succeed.

TIP RANKS


About GeoVax
GeoVax Labs, Inc. is a clinical-stage biotechnology company developing novel vaccines against infectious diseases and therapies for solid tumors. The Company’s lead clinical program is GEO-CM04S1, a next-generation COVID-19 vaccine currently in three Phase 2 clinical trials, being evaluated as (1) a primary vaccine for immunocompromised patients such as those suffering from hematologic cancers and other patient populations for whom the current authorized COVID-19 vaccines are insufficient, (2) a booster vaccine in patients with chronic lymphocytic leukemia (CLL) and (3) a more robust, durable COVID-19 booster among healthy patients who previously received mRNA vaccines. In oncology, the lead clinical program is evaluating a novel oncolytic solid tumor gene-directed therapy, Gedeptin®, having recently completed a multicenter Phase 1/2 clinical trial for advanced head and neck cancers. GeoVax is also developing a vaccine targeting Mpox and smallpox and, based on recent EMA regulatory guidance, anticipates progressing directly to a Phase 3 clinical evaluation, omitting Phase 1 and Phase 2 trials. GeoVax has a strong IP portfolio in support of its technologies and product candidates, holding worldwide rights for its technologies and products. For more information about the our clinical trials and other updates, visit our website: www.geovax.com.
Forward-Looking Statements
This release contains forward-looking statements regarding GeoVax’s business plans. The words “believe,” “look forward to,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax is able to obtain acceptable results from ongoing or future clinical trials of its investigational products, GeoVax’s immuno-oncology products and preventative vaccines can provoke the desired responses, and those products or vaccines can be used effectively, GeoVax’s viral vector technology adequately amplifies immune responses to cancer antigens, GeoVax can develop and manufacture its immuno-oncology products and preventative vaccines with the desired characteristics in a timely manner, GeoVax’s immuno-oncology products and preventative vaccines will be safe for human use, GeoVax’s vaccines will effectively prevent targeted infections in humans, GeoVax’s immuno-oncology products and preventative vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete development, there is development of competitive products that may be more effective or easier to use than GeoVax’s products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control.
Further information on our risk factors is contained in our periodic reports on Form 10-Q and Form 10-K that we have filed and will file with the SEC. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as may be required by law. GeoVax is a client of Institutional Analyst, the publisher of the Biotech Stock Review and has been retained with a combination of cash and equity for ongoing progress, news coverge and research reporting for five thousand dollars per month.
Company Contact:
info@geovax.com
678-384-7220
Media Contact:
Jessica Starman
media@geovax.com