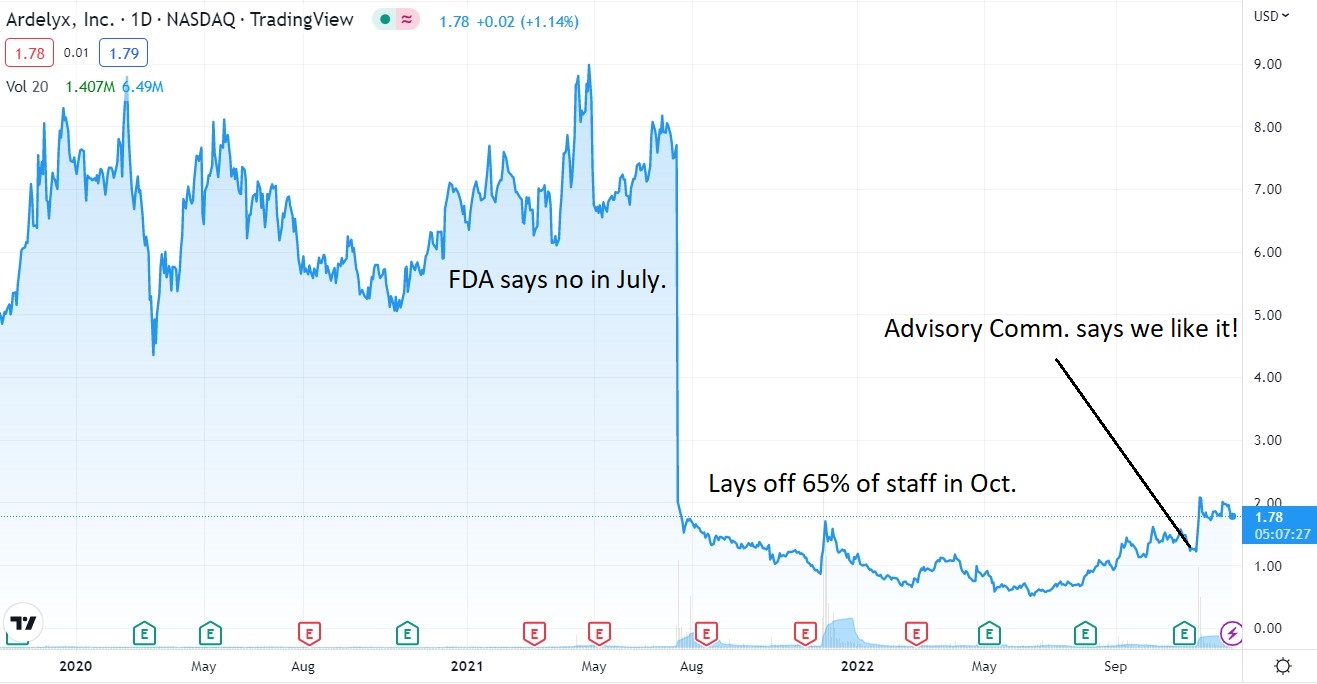
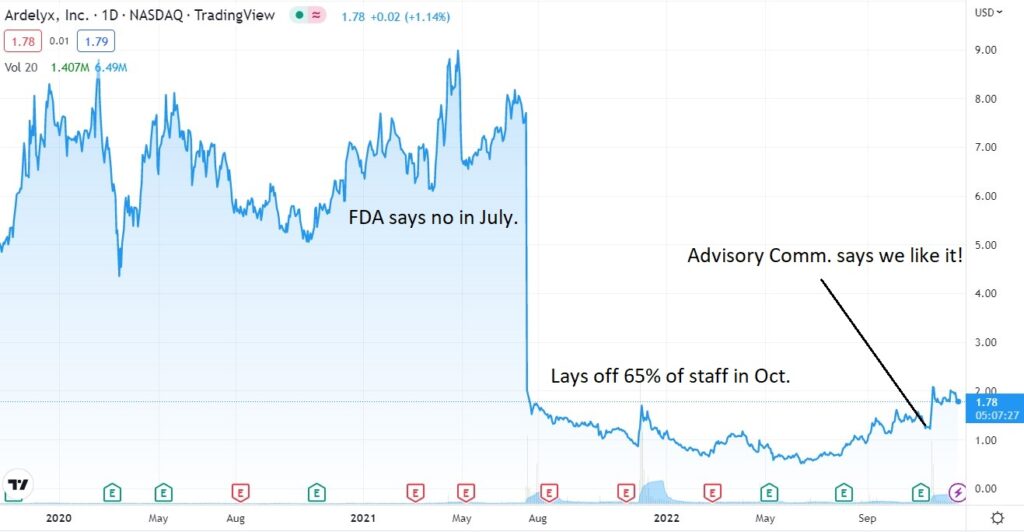
Oh boy, another day, another snag. There goes our Christmas bonus, dang.
It’s been a gut wrenching couple years for shareholders. Is it big payoff time or another beatdown, in the next of couple weeks. Time will tell.
Ardelyx Provides Update on FDA.
(Dec 16th) FDA says it needs a “few more” weeks, you know the holidays, New Years and all that..
Earlier today, Peter Stein , M.D., Director of the Office of New Drugs, Center for Drug Evaluation and Research of the U.S. Food and Drug Administration (FDA), requested a meeting with Ardelyx to discuss the status of his review of the appeal. After a productive conversation, Dr. Stein communicated that the FDA is finalizing the response and may require up to a few more weeks to do so. Ardelyx will communicate the response to the appeal once it has been received from the FDA.
As a reminder they failed in July of 2021, appeal was denied the following February and then low-and-behold, the FDA advisory committee say “we like it.”
RELATED (NOV): Adding Ardelyx (ARDX) $1.57 to Watch List.
SIGNIFICANT HEADLINES
2022
- (DEC 16TH) Ardelyx Provides Update on FDA Appeal
- (NOV) Ardelyx Announces FDA Advisory Committee Votes that the Benefits of XPHOZAH® (tenapanor) Outweigh its Risks
- (NOV) National Kidney Foundation Survey Finds Dialysis Patients Struggle to Comply With Pill Regimen.
- (JUNE) HealthCare Royalty Partners Will Invest $20 Million Post FDA Approval.
- (APRIL) Ardelyx Announces FDA Advisory Committee Meeting. Scheduled for November 16h.
- (FEB) SLR Capital Partners Invests $27.5 Million.
2021 - (NOV) Ardelyx to Pursue Formal Dispute Resolution with FDA
- (JULY) FDA Says Denied – Back to the Drawing Board?
DISCLAIMERS
About Ardelyx, Inc.
Ardelyx was founded with a mission to discover, develop and commercialize innovative first-in-class medicines that meet significant unmet medical needs. Ardelyx’s first approved product, IBSRELA ® (tenapanor) is available in the United States and Canada . Ardelyx is developing XPHOZAH ® (tenapanor), a novel product candidate to control serum phosphorus in adult patients with CKD on dialysis, which has completed three successful Phase 3 trials. Ardelyx has a Phase 2 potassium lowering compound, RDX013, for the potential treatment of elevated serum potassium, or hyperkalemia, a problem among certain patients with kidney and/or heart disease and an early-stage program in metabolic acidosis, a serious electrolyte disorder in patients with CKD. Ardelyx has established agreements with Kyowa Kirin in Japan , Fosun Pharma in China and Knight Therapeutics in Canada for the development and commercialization of tenapanor in their respective territories. For more information, please visit https://ardelyx.com/ and connect with us on Twitter @Ardelyx, LinkedIn and Facebook .
Forward Looking Statements
To the extent that statements contained in this press release are not descriptions of historical facts regarding Ardelyx, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor of the Private Securities Reform Act of 1995, including Ardelyx’s current expectation regarding the timing of the OND’s decision on its appeal. Such forward-looking statements involve substantial risks and uncertainties that could cause Ardelyx’s future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, uncertainties associated with the regulatory approval process. Ardelyx undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to Ardelyx’s business in general, please refer to Ardelyx’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 3, 2022 , and its future current and periodic reports to be filed with the Securities and Exchange Commission.