DALRADA aims to introduce its VIA Test kit to females in India – of which there is 497 million.
In 2012, the U.S. Preventive Services Task Force, American College of Obstetricians and Gynecologists and American Cancer Society recommended that women, beginning at age 21, should start Pap test screening every three years.
Unfortunately, cervical cancer screening is not always available. This is particularly true in many developing countries, and in areas with few financial resources for medical testing and treatment. Pap smears are considered the gold standard of cervical screening. Unfortunately, they require skilled practitioners and good laboratories to be effective.
That is why doctors have developed a test known as visual inspection with acetic acid (Vinegar) or the VIA test. It is cheap, easy, and effective.
In 2016, 22 million pap smears were performed in the United States alone.
In India there are 497 million females, making for a large addressable market for Dalrada’s VIA test kit. Of course, the number reduces down with age restrictions and other variables, but that’s still a huge number. Even at $5 a kit, we’re talking serious potential!
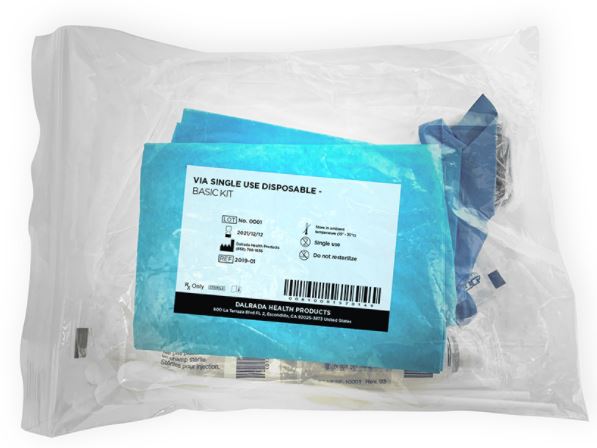
VIA TEST KIT
The basic Dalrada Visual Inspection with Acetic Acid (VIA) has been used in clinical trials in India and so far, the findings are positive – for patients and practitioners alike.
Drawing on 20+ years of obstetrics and gynecology clinical and surgical experience, Dalrada’s kit is designed to be accessible, affordable, and of proven quality.
The kit is based on the World Health Organization’s recommended procedure for conducting visual inspection with acetic acid (VIA).
The kit makes it easy for screening centers to manage multiple aspects such as ensuring consistency and quality of the kit’s components as well as confirming that all active ingredients are sourced from FDA or ISO-approved facilities. This also ensures the validation process is consistent, as each kit uses identical components.
Long-term studies have been conducted around the world that ultimately show the effectiveness of the VIA-based screening approach. In India, for example, cervical cancer mortality was reduced by 31% over a period of 15 years among women screened with VIA.
In Ghana, the incremental economic costs per woman screened with VIA ranged from $4.93 and $14.75.
RELATED: Visual Inspection With Acetic Acid (VIA) as a Low Cost Cervical Screen
Cervical Cancer Study Compares Dalrada Health VIA Screening Kits To Pap Smear, Peer Reviewed Journal – The Gynecologist – Publishes Findings.
SAN DIEGO , Oct. 26, 2020 /PRNewswire/ — Dalrada Financial Corp. (OTC Pink: DFCO) announces The Gynecologist, an international peer reviewed open access scholarly journal, has published clinical study research results on Visual Inspection with Acetic Acid (VIA) using Dalrada Health’s VIA inspection kits compared to Pap smear, a traditional cervical cancer screening method. The study titled “Cervical Cancer Screening with Pap Smear and VIA Using a Novel VIA Kit in a Tertiary Care Hospital in South India” was conducted by Physician-Scientist, Dr. Payal Keswarpu , Dalrada Health’s Chief Medical Officer, Westchester Knowledge Works in Bangalore, India, and Hethyshi Ranganna from the Department of Obstetrics and Gynecology, Kempegowda Institute of Medical Sciences in Bangalore, India .
Use of Dalrada Health’s patent pending VIA kits by a healthcare professional in a primary healthcare setting simplifies procedures for rapid management of multiple aspects to early detection of cervical cancer. Composed of components manufactured in FDA or ISO-approved facilities, Dalrada Health’s VIA kits ensure quality control for consistent validation processes. Due to the pandemic, the study was severely delayed, however due to the efforts of Dr. Payal and Dalrada Health team, the company was able to complete the last remaining patient tests.
The study compared Pap smear to Dalrada Health’s Visual Inspection with Acetic Acid (VIA) Kit for cervical cancer detection. In the 100-person study , participants ranged in age from 30-65 years-old. Study results include:
- More than 94% of the time, VIA and Pap tests results concurred
- VIA was highly specific in comparison to Pap smear
- Biopsy can be immediately taken in VIA positive cases
- Ready-to-use kits will aid automated imaging analysis in the future
- Detecting and timely treatment of latent infections is an added benefit of VIA kits
Cervical cancer is one among the top three cancers affecting women younger than 45 years in 146 of 185 countries assessed by the World Health Organization . Additional statistics on cervical cancer indicate:
- One woman dies ever two minutes from cervical cancer
- More than 75% of first-time screened women are diagnosed in advanced stages cervical cancer
- 85% of cervical cancer deaths occur in low-and middle-income countries (WHO)
- VIA screenings reduced cervical cancer mortality by 31% over a 15-year period ( India )
Further studies indicate that VIA screening, implemented by trained healthcare professionals with products of sustained quality assurance, is an effective method for cervical cancer screening in countries that have large low-to-medium income populations.
Brian Bonar , CEO of Dalrada, states “Reducing the high mortality rate from cervical cancer is being addressed by Dalrada Health. Through a comprehensive approach with primary prevention, effective screening, and treatment programs, early detection of cervical cancer can save countless lives around the world. Dalrada Health’s goal is to provide affordable healthcare resources to reverse the devastating effect of cervical cancer globally.”
Based on the World Health Organization’s recommended procedure for conducting visual inspection with acetic acid, Dalrada Health’s VIA kits contain all the necessary elements required for the procedure to be conducted by a healthcare professional in a primary healthcare setting. For additional information, visit https://dalradahealth.com
About Dalrada Health
A wholly-owned subsidiary of Dalrada Financial Corp., Dalrada Health is focused on solving health problems around the world. The company develops products and services that address the unmet needs of consumers due to accessibility, affordability, or availability. With operations in the U.S., Malaysia, and India, Dalrada Health can reach underserved markets through strong partnerships and the development of efficient supply chains. Dalrada Health is committed to solving universal health problems through improved methodologies resulting in products and services that benefit the global marketplace. For details, visit www.dalradahealth.com
About Dalrada (DFCO)
Dalrada Financial Corp. (OTC Pink: DFCO ) solves real-world problems by producing innovation-focused and technologically centered solutions on a global level. Delivering next-generation manufacturing, engineering, and healthcare products and services designed to propel growth, Dalrada is a team of industry experts and an organization built upon a strong foundation of financial capital. The Company and its subsidiaries are positioned for stable long-term growth through intelligent market research, sound business acumen, and established operational infrastructure. For more information, visit www.dalrada.com or call 1-858-283-1253.
Disclaimer
Statements in this press release that are not historical facts are forward-looking statements, including statements regarding future revenues and sales projections, plans for future financing, the ability to meet operational milestones, marketing arrangements and plans, and shipments to and regulatory approvals in international markets. Such statements reflect management’s current views, are based on certain assumptions and involve risks and uncertainties. Actual results, events, or performance may differ materially from the above forward-looking statements due to a number of important factors, and will be dependent upon a variety of factors, including, but not limited to, our ability to obtain additional financing that will allow us to continue our current and future operations and whether demand for our products and services in domestic and international markets will continue to expand. The Company undertakes no obligation to publicly update these forward-looking statements to reflect events or circumstances that occur after the date hereof or to reflect any change in the Company’s expectations with regard to these forward-looking statements or the occurrence of unanticipated events. Factors that may impact the Company’s success are more fully disclosed in the Company’s most recent public filings with the U.S. Securities and Exchange Commission (“SEC”), including its annual report on Form 10-K.
![]() View original content to download multimedia: http://www.prnewswire.com/news-releases/cervical-cancer-study-compares-dalrada-health-via-screening-kits-to-pap-smear-peer-reviewed-journal—the-gynecologist–publishes-findings-301159694.html
View original content to download multimedia: http://www.prnewswire.com/news-releases/cervical-cancer-study-compares-dalrada-health-via-screening-kits-to-pap-smear-peer-reviewed-journal—the-gynecologist–publishes-findings-301159694.html
SOURCE Dalrada Financial Corp.