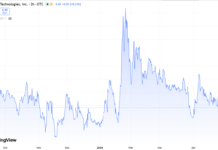
Adding ImmunityBio (IBRX) $2.40 to Watch List.
You never know where you’ll find a great idea (potentially great that is). How’s Megyn Kelly for a source! It’s of course not a financial podcast, which is probably a good thing. It makes the technology claims seem a bit more believable, without a money-making angle.

(See interview below)
Though there is nothing wrong with profiting from saving lives or making lives better, and/or illness free – mind you. We’ve been doing it for decades.
Anyway in the spectacular interview with Dr. Patrick Soon-Shiong, he chats about Interluken 15 ‘IL-15’ and he states, “I didn’t invent this, God invented this.” We like the sounds of that. Always good to have the big man on your side, in biotech stock infested waters!
We’re just putting this interview out for now, with a couple links below that are Wall Street related – as in how do I make money from this idea.
We met the Doctor back in 2000 or 2001, during a investor roadshow for American Pharmaceutical (APP) which was going public. Since then he’s been pulling rabbits out of his hat ever decade. While APP was based in Chicago, the Doctor is in reality based in Los Angeles and he has made billions, and now owns stakes in the LA Dodgers and the LA Times.
So to make a long story short, he’s neither a quack nor Biotech stock hustler. Listen to the interview.
While only time will tell, despite a $2 billion market cap, this rabbit has retreated far deep into the hat. One man’s loss can equal another’s opportunity. The stock is down from $30..so far.

INTERVIEW WITH MEGYN KELLY
UNRELATED: GeoVax Labs, Inc. (GOVX) Q2 2025 Earnings Call Transcript
ACTIVE TRADER

RECENT NEWS
$80 MILLION RAISE
They probably upset the average retail investor, when they announced that they raised $80 million (cries of dilution) from two investors, Mike Rockefeller @ Woodline Master Fund and Martin Kobinger @ Heights Capital (via CVI). Especially since they already had $135 million in the bank.
But think of it this way. If these guys with sophisticated in house analysts – decided to write $80 million in checks, shouldn’t we be following in the footsteps? Ya think? Congrats to Piper Sandler & Co. for acting as the exclusive placement agent and ringing the money bell!
Interview With Mike Rockefeller on Bloomberg.

About ImmunityBio, Inc.
ImmunityBio is a vertically-integrated commercial stage biotechnology company developing next-generation therapies that bolster the natural immune system to defeat cancers and infectious diseases. The Company’s range of immunotherapy and cell therapy platforms, alone and together, act to drive and sustain an immune response with the goal of creating durable and safe protection against disease. Designated an FDA Breakthrough Therapy, ANKTIVA® is the first FDA-approved immunotherapy for non-muscle invasive bladder cancer CIS that activates natural killer cells, T cells, and memory T cells for a long-duration response. The Company is applying its science and platforms to treating cancers, including the development of potential cancer vaccines, as well as developing immunotherapies and cell therapies that we believe sharply reduce or eliminate the need for standard high-dose chemotherapy. These platforms and their associated product candidates are designed to be more effective, accessible, and easily administered than current standards of care in oncology and infectious diseases. For more information, visit ImmunityBio.com (Founder’s Vision) and connect with us on X (Twitter), Facebook, LinkedIn, and Instagram.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, such as statements regarding the timing and size of the proposed offering, the potential exercise of the warrants being offered and resulting additional proceeds to the Company, the anticipated closing of the equity financing described herein and use of proceeds to be received from such financing, the application of the Company’s science and platforms to treat cancers or develop cancer vaccines, immunotherapies and cell therapies that reduce or eliminate the need for standard high-dose chemotherapy. Statements in this press release that are not statements of historical fact are considered forward-looking statements, which are usually identified by the use of words such as “anticipates,” “believes,” “continues,” “goal,” “could,” “estimates,” “scheduled,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “indicate,” “projects,” “seeks,” “should,” “will,” “strategy,” and variations of such words or similar expressions. Statements of past performance, efforts, or results of our preclinical and clinical trials, about which inferences or assumptions may be made, can also be forward-looking statements and are not indicative of future performance or results. Forward-looking statements are neither forecasts, promises nor guarantees, and are based on the current beliefs of ImmunityBio’s management as well as assumptions made by and information currently available to ImmunityBio. Such information may be limited or incomplete, and ImmunityBio’s statements should not be read to indicate that it has conducted a thorough inquiry into, or review of, all potentially available relevant information. Such statements reflect the current views of ImmunityBio with respect to future events and are subject to known and unknown risks, including business, regulatory, economic and competitive risks, uncertainties, contingencies and assumptions about ImmunityBio, including, without limitation, (i) whether the equity financing transaction described herein will close on the timeline anticipated, if at all, (ii) those related to the regulatory submission, filing and review process and the timing thereof, (iii) the ability of ImmunityBio to fund its ongoing and anticipated clinical trials, (iv) whether clinical trials will result in registrational pathways, (v) whether clinical trial data will be accepted by regulatory agencies, (vi) the ability of ImmunityBio to continue its planned preclinical and clinical development of its development programs through itself and/or its investigators, and the timing and success of any such continued preclinical and clinical development, patient enrollment and planned regulatory submissions, (vii) potential delays in product availability, regulatory approvals, and reimbursement decisions, (viii) ImmunityBio’s ability to retain and hire key personnel, (ix) ImmunityBio’s ability to obtain additional financing to fund its operations and complete the development and commercialization of its various product candidates, (x) potential product shortages or manufacturing disruptions that may impact the availability and timing of product, (xi) ImmunityBio’s ability to successfully commercialize its approved product and product candidates, (xii) ImmunityBio’s ability to scale its manufacturing and commercial supply operations for its approved product and future approved products, and (xiii) ImmunityBio’s ability to obtain, maintain, protect, and enforce patent protection and other proprietary rights for its product candidates and technologies. More details about these and other risks that may impact ImmunityBio’s business are described under the heading “Risk Factors” in the Company’s Form 10-K filed with the U.S. Securities and Exchange Commission (SEC) on March 3, 2025, and the Company’s Form 10-Q filed with the SEC on May 12, 2025, and in subsequent filings made by ImmunityBio with the SEC, which are available on the SEC’s website at www.sec.gov. ImmunityBio cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date hereof. ImmunityBio does not undertake any duty to update any forward-looking statement or other information in this press release, except to the extent required by law. Not a client, but it can’t hurt to call.
Contacts
Investors
Hemanth Ramaprakash, PhD, MBA
ImmunityBio, Inc.
+1 858-746-9289
Hemanth.Ramaprakash@ImmunityBio.com
Media
Sarah Singleton
ImmunityBio
+1 415-290-8045
Sarah.Singleton@ImmunityBio.com