The Company stated yesterday that it “has received positive comments from the FDA in regard to the Company’s request to determine and agree on the Study’s potential new primary efficacy endpoints, including the rate of sustained clinical resolution of symptoms of COVID-19, which addresses the shift in COVID-19 clinical outcome observed over the course of the pandemic, and, therefore, to have more meaningful study endpoints for the FDA to consider for potential Emergency Use Authorization.”
Normally a headline “adjusting endpoints” makes our hair stand on end. It makes us feel like a Company doing their trial, is not meeting their endpoints. Logical right?
Not so, in this case, in our opinion. First, Revive is seeking to expand the endpoints, not reduce them. There is a lot of data collected during their trial beyond the original two data endpoints — did they have to go to hospital or die?
Meaning to us there may be a lot of potential ‘good’ things (benefits) from taking Bucillamine as well, in addition to reducing bad things from happening. Further meaning to us, that this is data the FDA should be aware of, and data patients who contract Covid should be aware of. Sure, being kept out of the hospital and/or preventing death is great – but what it can also reduce some of the other awful symptoms. Sounds like a win-win to us.
What kind of things? Primarily things caused by inflammation (plenty of decidedly bad things) starting with elevated temperature, lowered oxygen levels, increases in blood pressure and heart rate, cough, headache, catarrh, etc. As a reminder, Revive is seeking to combat these things, not by combatting the virus, but by combatting inflammation.
An investor on Reddit speculates that “Now with Omicron the game has changed, since hospitalization is lower, and symptoms are more severe. With symptoms as endpoints things change dramatically.”
Changing Endpoints from Hospitalization to Symptoms – What does this mean for Revive?
So, in summary, the more data the FDA has on the benefits of Bucillamine, the better.
Revive Therapeutics Provides Update on Phase 3 Clinical Trial for Bucillamine in COVID-19
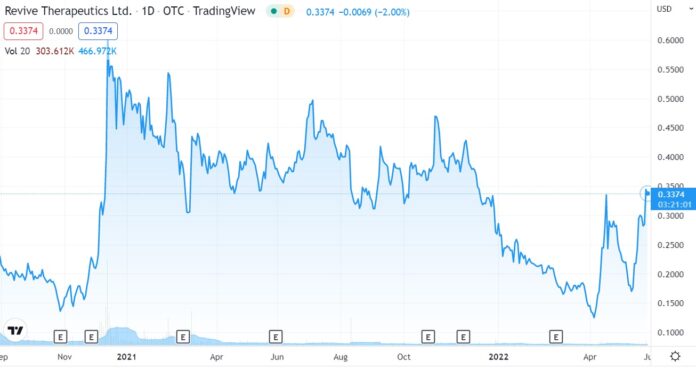
TORONTO, May 26, 2022 (GLOBE NEWSWIRE) — Revive Therapeutics Ltd. (“Revive” or the “Company”) (OTCQB: RVVTF) (CSE: RVV) (FRANKFURT: 31R), a specialty life sciences company focused on the research and development of therapeutics for medical needs and rare disorders, is pleased to provide an update on the Company’s U.S. Food & Drug Administration (“FDA”) Phase 3 clinical trial (the “Study”) ( NCT04504734 ) to evaluate the safety and efficacy of Bucillamine, an oral drug with anti-inflammatory and antiviral properties, in patients with mild to moderate COVID-19.
The Company has received positive comments from the FDA in regards to the Company’s request to determine and agree on the Study’s potential new primary efficacy endpoints, including the rate of sustained clinical resolution of symptoms of COVID-19, which addresses the shift in COVID-19 clinical outcome observed over the course of the pandemic, and, therefore, to have more meaningful study endpoints for the FDA to consider for potential Emergency Use Authorization. The FDA has agreed that the Company may unblind the pre-dose-selection data for the first 210 patients of the Study to further support the new primary endpoint. Before unblinding the pre-dose selection data for the first 210 patients, the Company will submit a Data Access Plan to the FDA in early June 2022 with the aim to unblind the pre-dose selection data and submit the amended Study protocol with the new primary efficacy endpoints to the FDA.
In addition, the Data Safety and Monitoring Board (“DSMB”) are scheduled to meet thereafter to evaluate the interim clinical and safety data and may make a recommendation on continuing the Study or advise on halting the Study early due to positive efficacy based on other clinical outcomes such as the rate of sustained clinical resolution of symptoms of COVID-19. The DSMB supported the continuation of the Study in its last meeting as there were no serious adverse events or safety concerns reported.
The Company believes that with the Omicron variant, including the BA.2 variant, being the dominant strain over the Delta variant, there is an urgent unmet need to treat symptom resolutions in addition to preventing hospitalizations.
The Company is not making any express or implied claims that its product has the ability to eliminate or cure COVID-19 (SARS-2 Coronavirus) at this time.
Disclaimer.
Cautionary Statement
This press release contains ‘forward-looking information’ within the meaning of applicable Canadian securities legislation. These statements relate to future events or future performance. The use of any of the words “could”, “intend”, “expect”, “believe”, “will”, “projected”, “estimated” and similar expressions and statements relating to matters that are not historical facts are intended to identify forward-looking information and are based on Revive’s current belief or assumptions as to the outcome and timing of such future events. Forward looking information in this press release includes information with respect to the Company’s cannabinoids, psychedelics and infectious diseases programs. Forward-looking information is based on reasonable assumptions that have been made by Revive at the date of the information and is subject to known and unknown risks, uncertainties, and other factors that may cause actual results or events to differ materially from those anticipated in the forward-looking information. Given these risks, uncertainties and assumptions, you should not unduly rely on these forward-looking statements. The forward-looking information contained in this press release is made as of the date hereof, and Revive is not obligated to update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as required by applicable securities laws. The foregoing statements expressly qualify any forward-looking information contained herein. Reference is made to the risk factors disclosed under the heading “Risk Factors” in the Company’s annual MD&A for the fiscal year ended June 30, 2021, which has been filed on SEDAR and is available under the Company’s profile at www.sedar.com Revive is a client of Institutional Analyst Inc and is compensated to provide news coverage and reporting. Please see report for full disclosure and disclaimer details.