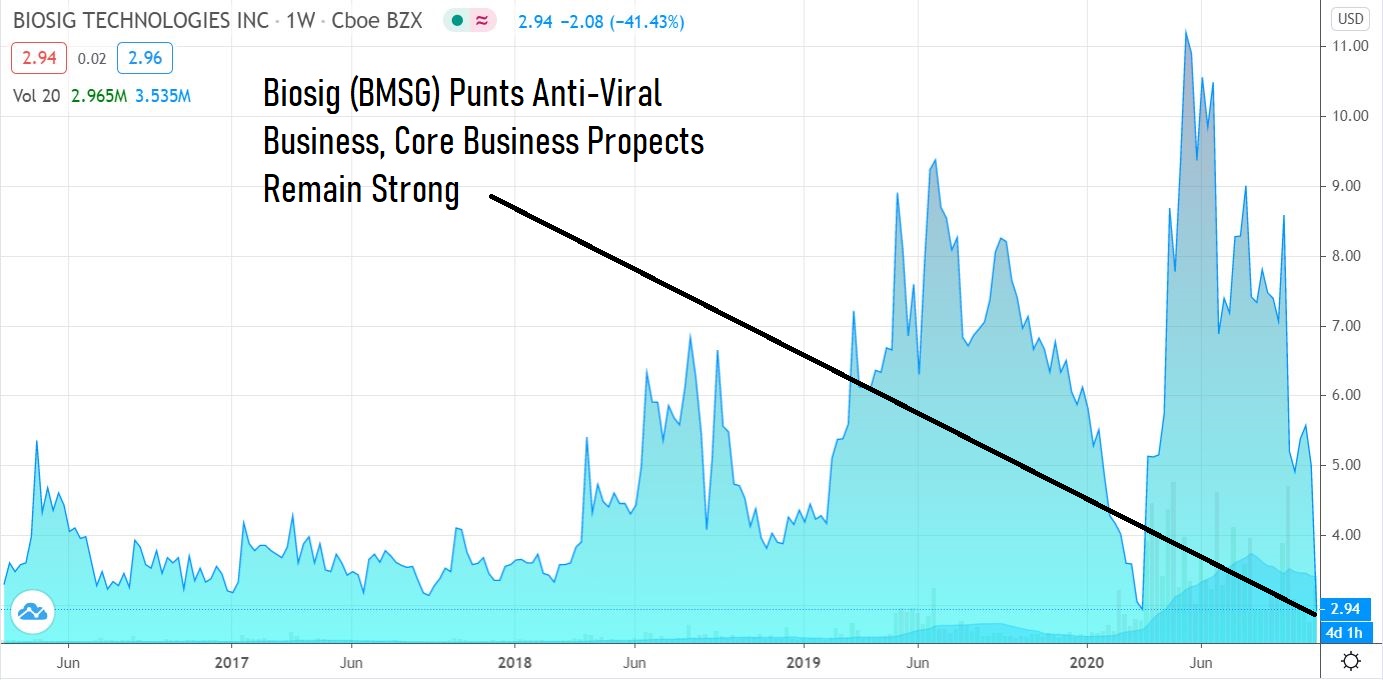
Tough setback for newly launched Anti-Viral Endeavor ViralClear, Back to the Business of PURE EP™.
We’ve been following this great little technology company since January. The Company later launched a CoronaVirus Anti-Viral Initiative (in March) which as of today, has been halted – which means putting their full focus back on their amazing heart monitoring technology called PURE EP™.
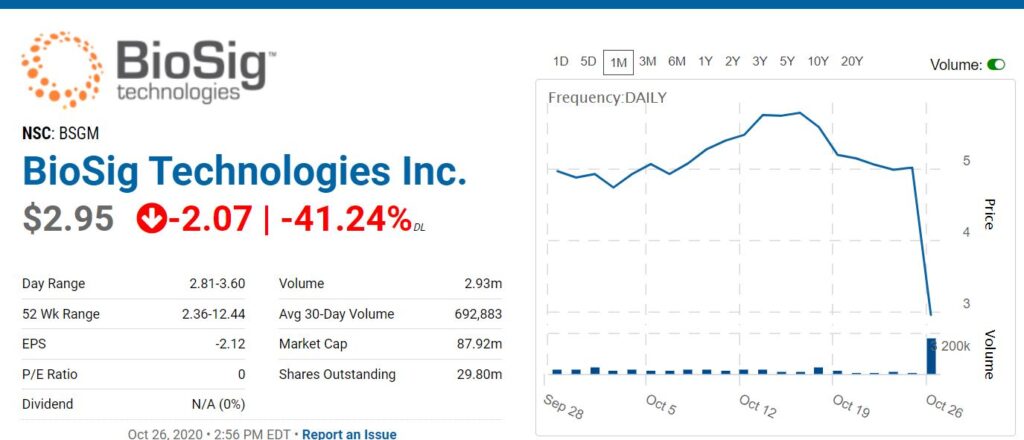
The share price today has been trading (by Corona traders) as if ViralClear was their only business – when that is far from the case.
The most common heart arrhythmia PURE EP™ can monitor during surgery is AFIB which affects 33.5 million people worldwide and 6.1 million in the United States. PURE EP™ could help doctors treat patients much faster and potentially reduce the need for repeat procedures.
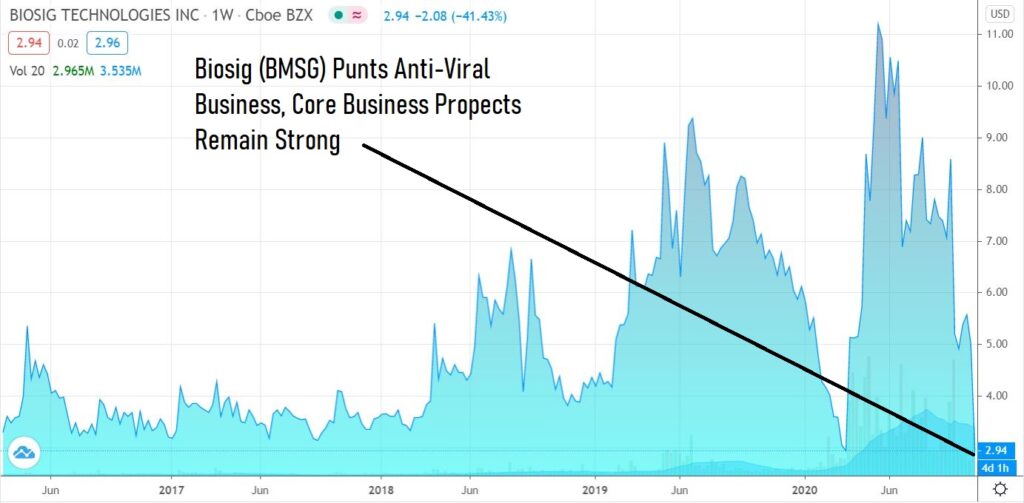
Though we have been following the Company for nearly a year, our attention was distracted (confused) over the acquisition and launch of an anti-viral initiative. We are studying in-depth now and adding to the Watch List. There is plenty of information related to the heart monitoring business, and we suggest readers visit the company website. It’s back to the business at hand and it is very exciting indeed.
FDA CLEARED PURE EP™

What will soon be ‘old’ news.
ViralClear halts its Phase 2 Hospitalized COVID-19 Trial
Westport, CT, Oct. 26, 2020 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (Nasdaq: BSGM) (“BioSig” or the “Company”) and its majority owned subsidiary, ViralClear Pharmaceuticals, Inc. (ViralClear), announced the halting of its signal finding Phase 2 trial, “A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study of the Efficacy and Safety of Oral Merimepodib in Combination with Intravenous Remdesivir in Adult Patients with Advanced Coronavirus Disease 2019 (COVID-19)”.
After the implementation of a protocol amendment that expanded the size of the trial from 40 to 80 hospitalized COVID-19 patients, and that limited enrollment to seriously ill patients, (NIAID Grade 3, who required high flow, high concentration oxygen to maintain adequate oxygenation) the Safety Monitoring Committee (SMC) was unblinded for safety reasons since these patients are at higher risk for dying from their disease. At the time of the most recent review of the data by the SMC, 44 patients had been enrolled in the trial of whom 42 had received study drug (either merimepodib solution or matching placebo). This most recent review of the data documented all 22 Grade 4 patients were discharged from the hospital and did not relapse during the 37 day follow-up period. However, patients who were NIAID Grade 3 patients (n = 20) at the time of enrollment had markedly different outcomes. Specifically, the unblinded SMC detected an imbalance in survival rates in these NIAID Grade 3 patients between the placebo and merimepodib making it unlikely that the trial would meet its primary safety endpoints. The company has therefore elected to stop enrollment into the clinical trial. Patients will be followed as per the protocol for safety monitoring; however, no further study drug treatments will be administered.
At this time, the Company does not intend to further develop merimepodib. However, the Company will see if other parties are interested in acquiring or licensing merimepodib.
About BioSig Technologies
BioSig Technologies is a medical technology company commercializing a proprietary biomedical signal processing platform designed to improve signal fidelity and uncover the full range of ECG and intra-cardiac signals ( www.biosig.com ).
The Company’s first product, PURE EP(tm) System is a computerized system intended for acquiring, digitizing, amplifying, filtering, measuring and calculating, displaying, recording and storing of electrocardiographic and intracardiac signals for patients undergoing electrophysiology (EP) procedures in an EP laboratory.
Forward-looking Statements
This press release contains “forward-looking statements.” Such statements may be preceded by the words “intends,” “may,” “will,” “plans,” “expects,” “anticipates,” “projects,” “predicts,” “estimates,” “aims,” “believes,” “hopes,” “potential” or similar words. Forward- looking statements are not guarantees of future performance, are based on certain assumptions and are subject to various known and unknown risks and uncertainties, many of which are beyond the Company’s control, and cannot be predicted or quantified and consequently, actual results may differ materially from those expressed or implied by such forward-looking statements. Such risks and uncertainties include, without limitation, risks and uncertainties associated with (i) the geographic, social and economic impact of COVID-19 on our ability to conduct our business and raise capital in the future when needed, (ii) our inability to manufacture our products and product candidates on a commercial scale on our own, or in collaboration with third parties; (iii) difficulties in obtaining financing on commercially reasonable terms; (iv) changes in the size and nature of our competition; (v) loss of one or more key executives or scientists; and (vi) difficulties in securing regulatory approval to market our products and product candidates. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company’s filings with the Securities and Exchange Commission (SEC), including the Company’s Annual Report on Form 10-K and its Quarterly Reports on Form 10-Q. Investors and security holders are urged to read these documents free of charge on the SEC’s website at http://www.sec.gov. The Company assumes no obligation to publicly update or revise its forward-looking statements as a result of new information, future events or otherwise.
Andrew Ballou BioSig Technologies, Inc. Vice President, Investor Relations 54 Wilton Road, 2nd floor Westport, CT 06880 aballou@biosigtech.com 203-409-5444, x133