May 2020.
Higher rates of serious illness and death from coronavirus SARS-CoV-2 (COVID-19) infection among older people and those who have comorbidities suggest that age- and disease-related biological processes make such individuals more sensitive to environmental stress factors including infectious agents like coronavirus SARS-CoV-2.
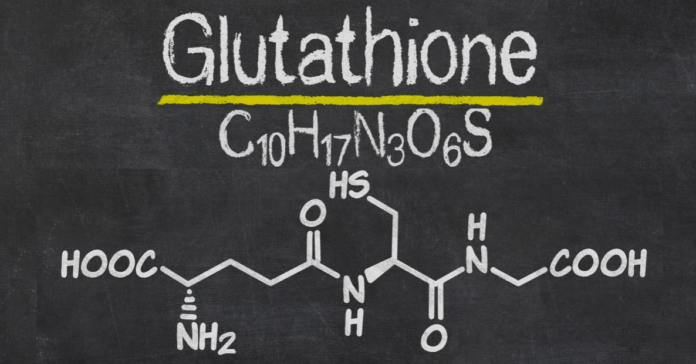
The hypothesis that glutathione deficiency is the most plausible explanation for serious manifestation and death in COVID-19 patients was proposed on the basis of an exhaustive literature analysis and observations. The hypothesis unravels the mysteries of epidemiological data on the risk factors determining serious manifestations of COVID-19 infection and the high risk of death and opens real opportunities for effective treatment and prevention of the disease.
NOTE
The overwhelming majority of people infected by SARS-CoV-2 have asymptomatic, mild, or moderate disease, and only 14% and 5% of patients developed severe or critical disease, respectively. (1) Higher rates of serious illness and death from COVID-19 infection among older people and those who have comorbidities suggest that age- and disease-related biological processes make such individuals more sensitive to environmental stress factors including infectious agents like coronavirus SARS-CoV-2.
Does Glutathione Deficiency Worsen COVID-19 Prognosis through the Risk Factors?
Numerous studies report that endogenous glutathione deficiency attributed to decreased biosynthesis and/or increased depletion of GSH represents a significant contributor to the pathogenesis of various diseases through mechanisms involving oxidative stress and inflammation.
Figure 1 summarizes the most common causes responsible for endogenous glutathione deficiency and also the mechanisms through which this deficiency may contribute to the pathogenesis of severe COVID-19 disease.

Age is a well-recognized risk factor for severe illness, complications, and death from COVID-19 infection. (8) Interestingly, animal and human studies indicate that the levels of endogenous glutathione progressively decline with aging, thereby making cells in the elderly more susceptible to oxidative damage caused by different environmental factors compared to younger individuals.Comorbidity is considered one of the major risk factors responsible for poor prognosis in COVID-19 patients. (5)
Interestingly, the deficiency in endogenous glutathione is common in individuals with chronic diseases as well as in individuals with a worsening prognosis of COVID-19. This means that decreased levels of glutathione occurring in COVID-19 patients with chronic diseases could be a triggering factor that shifts redox homeostasis toward oxidative stress, thereby exacerbating lung inflammation and leading to acute respiratory distress syndrome (ARDS), multiorgan failure, and death.
Sex-related COVID-19 mortality is one of the common epidemiologic findings around the globe, (9) suggesting sexual dimorphism in susceptibility to severe illness. It has been observed that men are significantly more likely to suffer severe effects of COVID-19 infection and experience a higher mortality rate that women. (9) In addition, men have lower plasma levels of reduced glutathione (GSH) than women, making men more susceptible to oxidative stress and inflammation.
Smoking is also considered a risk factor for severe complications and death from COVID-19. (10) Cigarette smoke is known to deplete the cellular glutathione pool in the airways, exacerbating oxidative damage and inflammation in the lung, which is likely the reason why smokers with COVID-19 more likely require intensive medical interventions.
Dietary factors may also contribute to endogenous glutathione deficiency in patients with severe COVID-19 illness. In particular, an insufficient consumption of fresh vegetables and fruits, natural sources of glutathione, seems to be an important but not yet established risk factor responsible for glutathione deficiency in patients with severe COVID-19 illness.
Thus, the relationship between risk factors and serious manifestations and death in COVID-19 patients could be attributable to a common cause, glutathione deficiency.
What Is the Primary Cause of Severe COVID-19 Illness: Glutathione or Vitamin D Deficiency?
The novel hypothesis that vitamin D (VD) deficiency is responsible for severe manifestations and death in COVID-19 patients has been proposed (11) and is actively being discussed by the scientific community. Several studies reported that glutathione levels positively correlate with active vitamin D. (12,13)
It has also been found that lower levels of l-cysteine (a rate-limiting precursor of GSH) and GSH correlated with lower vitamin D binding protein (VDBP) and VD levels in T2D patients (14) . l-cysteine supplementation is known to improve GSH status through upregulation of expression of VDBP, vitamin D 25-hydroxylase, and vitamin D receptor, thereby increasing vitamin D levels and decreasing inflammatory biomarkers in diabetic rats. (15)
Interestingly, a recent experimental study (16) showed that GSH deficiency and the associated increased oxidative stress epigenetically alters vitamin D regulatory genes and, as a result, the suppressed gene expression decreases VD biosynthesis, ultimately leading to a secondary deficiency of vitamin D.
It is important to note that the replenishment of GSH by l-cysteine treatment beneficially altered epigenetic enzymes methyltransferases and increased the expression of VD-metabolism genes.
This study provides important information that glutathione is essential for the control of endogenous vitamin D biosynthesis and demonstrates potential benefits of GSH treatment in reducing the deficiency of vitamin D. Taken together, these findings suggest that glutathione deficiency rather than vitamin D deficiency is a primary cause underlying biochemical abnormalities, including the decreased biosynthesis of vitamin D, and is responsible for serious manifestations and death in COVID-19 patients.
Antiviral, Anti-Inflammatory, and Anticoagulant Properties of Glutathione
ARTICLE SECTIONSJump To
Several studies indicate that higher levels of glutathione may improve an individual’s responsiveness to viral infections. In particular, glutathione is known to protect host immune cells through its antioxidant mechanism and is also responsible for optimal functioning of a variety of cells that are part of the immune system.
It is important to note that there is evidence that glutathione inhibits replication of various viruses at different stages of the viral life cycle (Figure 1), and this antiviral property of GSH seems to prevent increased viral loads and the subsequent massive release of inflammatory cells into the lung (“cytokine storm”).
The antiviral activity of glutathione was demonstrated in a study of De Flora et al. (17) who showed that a 6 month preventive administration of N-acetylcysteine (NAC, glutathione precursor) significantly reduced the incidence of clinically apparent influenza and influenza-like episodes, especially in elderly high-risk individuals.
In addition, pathophysiological conditions such as lung cell injury and inflammation in patients with severe ARDS were identified as the targets of NAC treatment. In particular, the deficiency of reduced glutathione in the alveolar fluid in ARDS patients was found to enhance lung cell injury by ROS/oxidative stress and inflammation, and this damage could be effectively prevented and treated by the administration of NAC (Figure 1).
Glutathione deficiency could also promote the increased activation of von Willebrand Factor causing coagulopathy in COVID-19 patients.
Glutathione Deficiency Exacerbates COVID-19 Illness: My Observations
Kursk State Medical University has been involved in a project on genetics of redox homeostasis in type 2 diabetes (T2D) since December, 2016. In April 2020, four patients from the control group were confirmed to have COVID-19. Blood samples were collected from the patients and used to measure total plasma ROS and GSH levels immediately after blood sampling. All four patients were females, nonsmokers, and without chronic diseases. Table 1 shows a description of the cases.
Patients with moderate and severe COVID-19 illness had lower levels of glutathione and higher ROS and ROS/GSH ratio in plasma than patients with mild disease, clearly indicating glutathione deficiency and oxidative stress signs in patients with serious disease manifestations.
Notably, only the patient with severe illness and a marked glutathione decrease is still severely sick, whereas the other patients with high/moderate levels of GSH have recovered.Table 1. Clinical and Laboratory Characteristics of Four COVID-19 Patientsa
| cases | disease severity | BMI, kg/m2/family history (FH) | day of clinical onset after contact with infected patient | clinical symptoms | day when symptoms disappeared | parameters of redox status, μmol/Lb |
|---|---|---|---|---|---|---|
| 1. female M. (age 34) | mild | 23.8 | 8 | fever 38 °C, mild myalgia | 6 | GSH, 0.712; ROS, 2.075; ROS/GSH ratio, 2.9 |
| 2. female P. (age 47) | mild | 21.0 | 10 | fever 37.3 °C, mild fatigue | 4 | GSH, 0.933; ROS, 1.143; ROS/GSH ratio, 1.2 |
| 3. female C. (age 44) | severe | 22.5, FH for diabetes | 4 | daily fever between 37.1 and 38.5 °C, dry cough, hoarseness, significant myalgia and fatigue (radiographic findings of pneumonia) | still sick, 24th day of illness (03.05.2020) | GSH, 0.079 (!); ROS, 2.73; ROS/GSH ratio, 34.6 (!) |
| 4. female R. (age 56) | moderate-to-severe | 33.0, FH for diabetes | 7 | fever up to 39 °C, a severe dry cough, dyspnea, significant fatigue and tachycardia (radiographic findings of pneumonia) | 16 | GSH, 0.531; ROS, 3.677 (!); ROS/GSH ratio, 6.9 (!) |
a
All four cases were nonsmokers without a history of chronic diseases. COVID-19 infection was confirmed by a PCR test in all cases.b
The parameters were measured 2 months before the patients became infected with coronavirus SARS-CoV-2.
Hypothesis
Endogenous glutathione deficiency appears to be a crucial factor enhancing SARS-CoV-2-induced oxidative damage of the lung and, as a result, leads to serious manifestations, such as acute respiratory distress syndrome, multiorgan failure, and death in COVID-19 patients.
When the antiviral activity of GSH is taken into account, individuals with glutathione deficiency seem to have a higher susceptibility for uncontrolled replication of SARS-CoV-2 virus and thereby suffer from an increasing viral load.
The severity of clinical manifestations in COVID-19 patients is apparently determined by the degree of impaired redox homeostasis attributable to the deficiency of reduced glutathione and increased ROS production. This assumption can be supported by our findings.
In particular, COVID-19 patients with moderate and severe illness had lower levels of glutathione, higher ROS levels, and greater redox status (ROS/GSH ratio) than COVID-19 patients with a mild illness.
Long-term and severe manifestations of COVID-19 infection in one of our patients with marked glutathione deficiency suggest that the degree of glutathione decrease correlates negatively with viral replication rate and that an increasing viral load exacerbates oxidative damage of the lung.
This finding suggests that the virus cannot actively replicate at higher levels of cellular glutathione, and therefore, milder clinical symptoms are observed with lower viral loads.
Glutathione deficiency is an acquired condition attributable to decreased biosynthesis and/or increased depletion of the endogenous GSH pool influenced by risk factors such as aging, male sex, comorbidity, and smoking alone or in combinations.
Glutathione deficiency in COVID-19 patients with serious illness may also be a result of decreased consumption of fresh vegetables and fruits (especially during winter and spring seasons), which contributes to over 50% of dietary glutathione intake. The hypothesis suggests that SARS-CoV-2 virus poses a danger only for people with endogenous glutathione deficiency, regardless which of the factors aging, chronic disease comorbidity, smoking or some others were responsible for this deficit.
The hypothesis provides novel insights into the etiology and mechanisms responsible for serious manifestations of COVID-19 infection and justifies promising opportunities for effective treatment and prevention of the illness through glutathione recovering with N-acetylcysteine and reduced glutathione.
Since the antiviral effect of glutathione is nonspecific, there is reason to believe that glutathione is also active against SARS-CoV-2. Therefore, restoration of glutathione levels in COVID-19 patients would be a promising approach for the management of the novel coronavirus SARS-CoV-2. Notably, long-term oral administration of N-acetylcysteine has already been tested as an effective preventive measure against respiratory viral infections. (1)
N-Acetylcysteine is widely available, safe, and cheap and could be used in an “off-label” manner. Moreover, parenteral injection of NAC or reduced glutathione (GSH is more bioavailable than NAC) could be an efficient therapy for COVID-19 patients with serious illness.
Horowitz et al. (18) just published a paper confirming this hypothesis: the authors reported the efficacy of glutathione therapy in relieving dyspnea associated with COVID-19 pneumonia. Nonetheless, the proposed hypothesis has to be confirmed in larger epidemiological and experimental studies, and also, clinical trials are needed to objectively assess the efficacy of N-acetylcysteine and reduced glutathione for the treatment and prevention of COVID-19 infection.
Author Information
I am grateful to my colleague Dr. Iuliia Azarova at Kursk State Medical University for sharing clinical and laboratory data of the patients with me.
- Corresponding Author
- Alexey Polonikov – Department of Biology, Medical Genetics and Ecology and Research Institute for Genetic and Molecular Epidemiology, Kursk State Medical University, 3 Karl Marx Street, 305041 Kursk, Russian Federation;
 http://orcid.org/0000-0001-6280-247X; Email: [email protected]
http://orcid.org/0000-0001-6280-247X; Email: [email protected]
- Alexey Polonikov – Department of Biology, Medical Genetics and Ecology and Research Institute for Genetic and Molecular Epidemiology, Kursk State Medical University, 3 Karl Marx Street, 305041 Kursk, Russian Federation;
- Notes The author declares no competing financial interest.
Acknowledgments
References
This article references 18 other publications.
- 1Wu, Z. and McGoogan, J. M. (2020) Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 323, 1239, DOI: 10.1001/jama.2020.2648 [Crossref], [PubMed], [CAS], Google Scholar
- 2Betteridge, D. J. (2000) What is oxidative stress?. Metab., Clin. Exp. 49 (2 Suppl 1), 3– 8, DOI: 10.1016/S0026-0495(00)80077-3 [Crossref], [PubMed], [CAS], Google Scholar
- 3Hekimi, S., Lapointe, J., and Wen, Y. (2011) Taking a ″good″ look at free radicals in the aging process. Trends Cell Biol. 21, 569– 576, DOI: 10.1016/j.tcb.2011.06.008 [Crossref], [PubMed], [CAS], Google Scholar
- 4Pisoschi, A. M. and Pop, A. (2015) The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 97, 55– 74, DOI: 10.1016/j.ejmech.2015.04.040 [Crossref], [PubMed], [CAS], Google Scholar
- 5Yang, J., Zheng, Y., Gou, X., Pu, K., Chen, Z., Guo, Q., Ji, R., Wang, H., Wang, Y., and Zhou, Y. (2020) Prevalence of comorbidities and its effects in coronavirus disease 2019 patients: A systematic review and meta-analysis. Int. J. Infect. Dis. 94, 91– 95, DOI: 10.1016/j.ijid.2020.03.017 [Crossref], [PubMed], [CAS], Google Scholar
- 6Lee, C. (2018) Therapeutic Modulation of Virus-Induced Oxidative Stress via the Nrf2-Dependent Antioxidative Pathway. Oxid. Med. Cell. Longevity 2018, 6208067, DOI: 10.1155/2018/6208067 [Crossref], [PubMed], [CAS], Google Scholar
- 7Forman, H. J., Zhang, H., and Rinna, A. (2009) Glutathione: overview of its protective roles, measurement, and biosynthesis. Mol. Aspects Med. 30, 1– 12, DOI: 10.1016/j.mam.2008.08.006 [Crossref], [PubMed], [CAS], Google Scholar
- 8Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., Xiang, J., Wang, Y., Song, B., Gu, X., Guan, L., Wei, Y., Li, H., Wu, X., Xu, J., Tu, S., Zhang, Y., Chen, H., and Cao, B. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395, 1054– 1062, DOI: 10.1016/S0140-6736(20)30566-3 [Crossref], [PubMed], [CAS], Google Scholar
- 9Borges do Nascimento, I. J., Cacic, N., Abdulazeem, H. M., von Groote, T. C., Jayarajah, U., Weerasekara, I., Esfahani, M. A., Civile, V. T., Marusic, A., Jeroncic, A., Carvas Junior, N., Pericic, T. P., Zakarija-Grkovic, I., Meirelles Guimarães, S. M., Luigi Bragazzi, N., Bjorklund, M., Sofi-Mahmudi, A., Altujjar, M., Tian, M., Arcani, D. M. C., O’Mathúna, D. P., and Marcolino, M. S. (2020) Novel Coronavirus Infection (COVID-19) in Humans: A Scoping Review and Meta-Analysis. J. Clin. Med. 9, 941, DOI: 10.3390/jcm9040941 [Crossref], Google Scholar
- 10Vardavas, C. I. and Nikitara, K. (2020) COVID-19 and smoking: A systematic review of the evidence. Tob. Induced Dis. 18, 20, DOI: 10.18332/tid/119324 [Crossref], [PubMed], [CAS], Google Scholar
- 11Grant, W. B., Lahore, H., McDonnell, S. L., Baggerly, C. A., French, C. B., Aliano, J. L., and Bhattoa, H. P. (2020) Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 12, 988, DOI: 10.3390/nu12040988 [Crossref], [CAS], Google Scholar
- 12Alvarez, J. A., Chowdhury, R., Jones, D. P., Martin, G. S., Brigham, K. L., Binongo, J. N., Ziegler, T. R., and Tangpricha, V. (2014) Vitamin D status is independently associated with plasma glutathione and cysteine thiol/disulphide redox status in adults. Clin. Endocrinol. (Oxford, U. K.) 81, 458– 466, DOI: 10.1111/cen.12449 [Crossref], [PubMed], [CAS], Google Scholar
- 13Jain, S. K., Micinski, D., Huning, L., Kahlon, G., Bass, P. F., and Levine, S. N. (2014) Vitamin D and L-cysteine levels correlate positively with GSH and negatively with insulin resistance levels in the blood of type 2 diabetic patients. Eur. J. Clin. Nutr. 68, 1148– 1153, DOI: 10.1038/ejcn.2014.114 [Crossref], [PubMed], [CAS], Google Scholar
- 14Jain, S. K., Kahlon, G., Bass, P., Levine, S. N., and Warden, C. (2015) Can L-Cysteine and Vitamin D Rescue Vitamin D and Vitamin D Binding Protein Levels in Blood Plasma of African American Type 2 Diabetic Patients?. Antioxid. Redox Signaling 23, 688– 693, DOI: 10.1089/ars.2015.6320 [Crossref], [PubMed], [CAS], Google Scholar
- 15Jain, S. K., Kanikarla-Marie, P., Warden, C., and Micinski, D. (2016) L-cysteine supplementation upregulates glutathione (GSH) and vitamin D binding protein (VDBP) in hepatocytes cultured in high glucose and in vivo in liver, and increases blood levels of GSH, VDBP, and 25-hydroxy-vitamin D in Zucker diabetic fatty rats. Mol. Nutr. Food Res. 60, 1090– 1098, DOI: 10.1002/mnfr.201500667 [Crossref], [PubMed], [CAS], Google Scholar
- 16Parsanathan, R. and Jain, S. K. (2019) Glutathione deficiency induces epigenetic alterations of vitamin D metabolism genes in the livers of high-fat diet-fed obese mice. Sci. Rep. 9, 14784, DOI: 10.1038/s41598-019-51377-5 [Crossref], [PubMed], [CAS], Google Scholar
- 17De Flora, S., Grassi, C., and Carati, L. (1997) Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur. Respir. J. 10, 1535– 1541, DOI: 10.1183/09031936.97.10071535 [Crossref], [PubMed], [CAS], Google Scholar
- 18Horowitz, R. I., Freeman, P. R., and Bruzzese, J. (2020) Efficacy of glutathione therapy in relieving dyspnea associated with COVID-19 pneumonia: A report of 2 cases. Respir Med. Case Rep 30, 101063, DOI: 10.1016/j.rmcr.2020.101063 [Crossref], [PubMed], [CAS], Google Scholar
This article is cited by 96 publications.
#RVVTF, $RVVTF