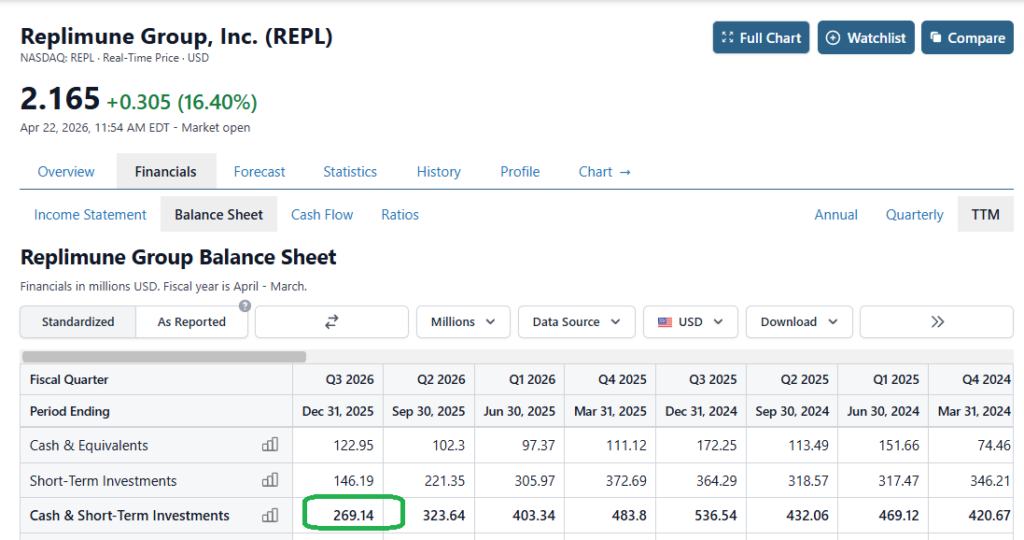
Here’s the balance sheet.. Imagine being the CEO?
Wife: “Honey, I’m going to the mall, do we have money in the checking?” CEO “Yes, but don’t go crazy, we’re down to $269 million and that has to last or we’ll be out of money by next Easter.”
Second CRL May Have Sealed RP1’s Fate – It’s Hard To See Positives
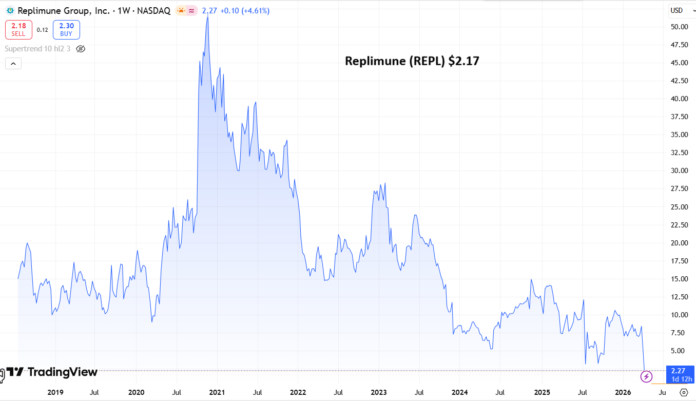
Edmund Ingham Apr 10, 2026, 3:30 PM Replimune Group, Inc. (REPL) Stock.
Share Price $4.76 (now $2.17)
Summary
Replimune Group, Inc. suffered a second FDA rejection for RP1 in anti-PD-1 failed melanoma, causing a sharp stock decline and casting doubt on its near-term prospects.
The FDA apparently cited inadequate evidence of effectiveness and trial design flaws, finding the IGNYTE study too heterogeneous and the confirmatory study insufficient.
REPL’s cash runway may not extend beyond 12 months, and the company may need to restructure or reprioritize resources, with RP2 in metastatic uveal melanoma as a possible focus.
Given persistent regulatory hurdles and uncertain funding, this does not appear to be a “buy the dip” opportunity for REPL.
News is breaking today that Replimune Group, Inc.’s (REPL) Biologics License Application (“BLA”) requesting approval for its drug candidate RP1 (vusolimogene oderparepvec) in the indication of anti-PD-1 failed melanoma has been rejected by the FDA – the second time the FDA has rejected the drug for commercial approval.
At the time of writing, Replimune stock has fallen in value by nearly 20%, trading at a value of $4.76 per share – trading is currently halted. Stock is down >50% year-to-date, and nearly 85% on a five-year basis.
In this note, I’ll provide some background and context to today’s news and speculate about what it means for the company and its share price and valuation going forward.
Investment Overview – Background To Today’s Decision
This is in fact my first time covering Replimune Group for Seeking Alpha, although I have frequently referenced the Woburn, Massachusetts-based biotech in my coverage of Iovance Biotherapeutics (IOVA) in the past.
As many readers will likely be aware, and as I noted in a March update on Iovance:
Iovance’s lifileucel, brand name Amtagvi, is a commercially available therapy for advanced melanoma, having been approved in February 2024, and together with Proleukin (aldesleukin), an interleukin-2, or IL-2, product used in the Amtagvi treatment regimen, helped Iovance post a revenue figure of $264m in 2025.
it took far longer than expected to secure approval for Amtagvi than initially thought – the original biologics license application (“BLA”) requesting FDA approval of the drug was submitted in 2022, but approval was not granted until 2024, testing Wall Street’s patience.
Replimune’s lead product is RP1, indicated for anti-PD-1 failed melanoma, i.e., patients did not respond to treatment with an immune checkpoint inhibitor – likely Merck’s (MRK) Keytruda (pembrolizumab), or Bristol-Myers Squibb’s (BMY) Opdivo (nivolumab).
Replimune, like Iovance, has undergone a long and tortuous path towards an FDA approval, however its journey has ultimately not been completed – as per its fiscal year Q3 10-Q filing from early February:
In November 2024, we announced submission of our first Biologics License Application, or BLA, to the U.S. Food and Drug Administration, or the FDA, for RP1 (vusolimogene oderparepvec) in combination with nivolumab for the treatment of adult patients with advanced melanoma who have previously received an anti-PD-1 containing regimen, and that the FDA has granted Breakthrough Therapy designation for RP1 in combination with nivolumab in the same setting.
The submission was made under the accelerated approval pathway. The FDA accepted our BLA and granted priority review with a Prescription Drug User Fee Act, or PDUFA, goal date of July 22, 2025. On July 21, 2025 the FDA issued a complete response letter, or CRL, for the RP1 BLA for the treatment of advanced melanoma.
The dreaded CRL outlined the FDA’s reasons for rejecting a application to approve a drug. Iovance notes:
The FDA stated in the CRL that it was unable to approve the application in its present form and that the IGNYTE trial was not considered to be an adequate and well-controlled clinical investigation that provided substantial evidence of effectiveness, including contribution of components. Furthermore, the FDA said the trial could not be adequately interpreted due to the heterogeneity of the patient population.
Clearly, Replimune was confident that it had addressed the FDA’s concerns – during a fireside chat at this year’s JP Morgan Healthcare conference in January, its CEO had told the audience:
“So near term, we’re looking forward to our approval, and we’re already ready to execute on our commercial launch plans. That involves working very closely with the oncology and interventional radiology stakeholders. We’ve done a lot of work on now and ready to go. And we’re also worked to simplify the logistics for our modalities so that we can enable next-day delivery and also stability at room temperature for our asset.”
In its press release announcing the FDA acceptance of its resubmitted BLA, Replimune describes the mechanism of action (“MoA”) of RP1 as follows:
RP1 (vusolimogene oderparepvec) is Replimune’s lead product candidate and is based on a proprietary strain of herpes simplex virus engineered and genetically armed with a fusogenic protein (GALV-GP R-) and GM-CSF, intended to maximize tumor killing potency, the immunogenicity of tumor cell death, and the activation of a systemic anti-tumor immune response.
It’s pivotal study used RP1 in combo with nivolumab – Bristol-Myers Squibb’s (BMY) PD-1 inhibitor Opdivo, and achieved an objective response rate of ~33%, however the FDA apparently concluded the study has enrolled too broad a patient population, making it hard to assess results, and was also unhappy with the design of a “confirmatory” study that Replimune intended to conduct post-approval, to confirm its Phase 1/2 data.
Replimune shares the following overview of IGNYTE study results as follows in its latest investor presentation:
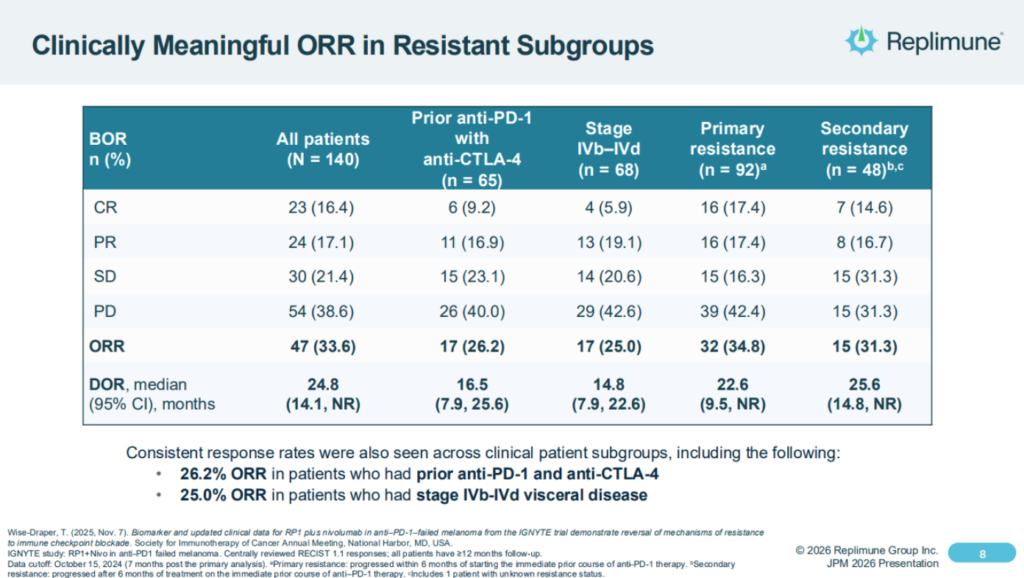
As the CEO explained back in January, the company had confidence that it was RP1, not the previously administered PD-1 therapy that was doing the “heavy lifting:
So one of the important questions you always get when you’re doing a single-arm trial, particularly with the combination is well, what is RP1 contributing? And I mentioned one of the ways we got to that was this very strict criteria so that we know that further PD-1-based treatments really wouldn’t do very much, as I said, about 5%. So when you see about a 34% response rate, we know that the RP1 is really contributing.
At the time of the first rejection, there was widespread surprise at the FDA’s rejection, with some blaming the new regime at the FDA – notably, Dr Vinay Prasad, the former Director of the Center for Biologics Evaluation and Research (“CBER”). With Prasad set to leave the FDA at the end of this month, however, it seems the FDA remains broadly unconvinced by Replimune’s data set, and does not believe the updated data and analytics provided since a Type A meeting last year materially changes the case for approval.
The reality is, oncolytic viruses have struggled to prove their mettle both in the clinic (RP1 formerly failed a study in cutaneous squamous cell carcinoma), and commercially – Amgen’s Imlygic, approved to treat melanoma in 2015, likely earns <$100m per annum (Amgen does not break out its revenue contribution but would likely do so if the figure was >$100m).
Replimune – What Happens Now?
Announcing its fiscal Q3 earnings in February this year, Replimune reported a net loss of $(247m), up from $(182m) in the prior year period, and a cash position of $269m.
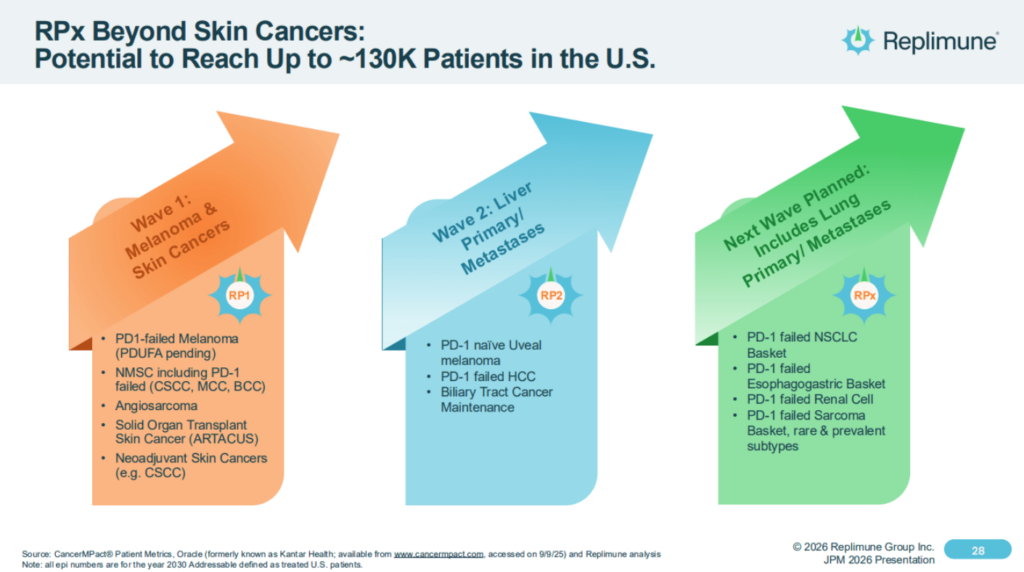
Replimune’s plans have always extended beyond approval in advanced melanoma to indications addressing ~130k cancer patients, as illustrated in the slide below:
Doubtless, Replimune management will feel hard done by today and will want to press ahead with studies, but seemingly, with a cash position of $269.1M and a quarterly cash burn of $70.9M, its funding runway may only extend into the first quarter of 2027. It’s likely Replimune would have looked to raise funds via a stock offering if its BLA had been approved today, at a much higher price than the current share price value of <$5, to fund a commercial launch, but it may now be challenging for the company to secure funding, or even issue debt.
In terms of the objective response rates (“ORRs”) and Complete Response Rates (“CRs”) observed in the IGNYTE study, they do seem broadly comparable to Amtagvi. However, Amtagvi seems to have performed well in the commercial setting – five-year analysis of the pivotal study of Amtagvi in melanoma patients revealed an ORR of 31.4%, a five-year overall survival (“OS”) of 19.7%, a median duration of response (“mDor”) of 36.5 months, and a median OS of 13.9 months.
Ultimately, however clinical studies are rarely comparable, patients have contrasting profiles, may respond differently to a particular therapy, and trial designs and outcomes must be crystal clear and pre-agreed with the regulatory authorities. Rightly or wrongly, the FDA was seemingly convinced by Iovance’s data submission, and unconvinced by Replimune’s.
At the time of writing, Replimune has not released any communications round the FDA’s decision – management will doubtless believe they had reached agreement with the administration on the data required to secure approval, and supplied it, but the final decision always rests with the administration (for better or worse).
April 10 Release post report
“It is deeply disappointing that the FDA has not exercised regulatory flexibility to meet patients’ needs given the data supporting strong efficacy and the favorable safety profile. Approximately 8,500 Americans with advanced melanoma die every year. The country’s foremost melanoma specialists stood behind the RP1 data. Patients and caregivers pleaded for urgency. All of it was met with inconsistent communication and a fragmented and slow-moving regulatory process which clearly puts U.S. innovation at risk,” said Sushil Patel, Ph.D., CEO of Replimune. “As we previously communicated, without timely accelerated approval, the development of RP1 will not be viable. We are devastated for our committed employees who have worked tirelessly for patients but at this point we have no choice but to eliminate jobs, including substantially scaling back our U.S. based manufacturing operations. A treatment desperately needed by patients will not be available. Not because the medicine failed. Because the system did.”
Dems fighting words!
As such, it is hard to know how the company will react. The company’s market cap has sunk to <$400m, and it may be the case Replimune will need to scale back operations and reprioritize resources – it would not necessarily be surprising to see a corporate restructuring, in order to reduce costs.
The post-confirmatory study in advanced melanoma is surely no longer required, and in many senses it feels as though Replimune is back at square one. The company does have a second program, RP2, currently progressing through a Phase 2/3 study in metastatic uveal melanoma, and I wonder if management will opt to prioritize this over RP1 going forward. Management says that:
The primary endpoints of the trial are overall survival and progression free survival, and key secondary endpoints are overall response rate and disease control rate. Phase 2/3 transition is expected in Q1 2027, with PFS analysis potentially supporting accelerated approval.
Concluding Thoughts – A Tough Day For Replimune, A Quick Turnaround In Fortunes Feels Unlikely
My take on today’s news is that the FDA has doubled down on the criticism of Replimune’s pivotal study it shared with its first CRL, and does not believe its criticisms have been addressed by the re-submission.
Facing its second rejection, my take would also be that Replimune cannot have too many complaints. Oncolytic viruses, while a promising potential therapy, have defied the best efforts of several pharmas and biotechs to devise an effective and safe therapy, and there did not seem to be enough signs that RP1 would have been a drastic improvement on Amtagvi, or potentially even PD-1 plus chemotherapy, while other melanoma therapies – Moderna’s (MRNA) / Merck’s (MRK) cancer “Vaccine” candidate for example, may soon provide another treatment option in a field of admittedly high unmet need.
For Replimune, its feels as though it is back to the drawing board, and management may need to take some tough decisions. It will certainly be interesting to see the company’s response, but whatever it may be, personally, and doubtless many readers will disagree, this does not feel like a “buy the dip” moment for Replimune stock.

If you are interested in keeping up to date with stocks making moves within the biotech, pharma and healthcare industries, and understanding the key trends and catalysts driving valuations ahead of the market, why not subscribe to my weekly newsletter via my Investing Group, Haggerston BioHealth?