It’s a bit of a stretch...
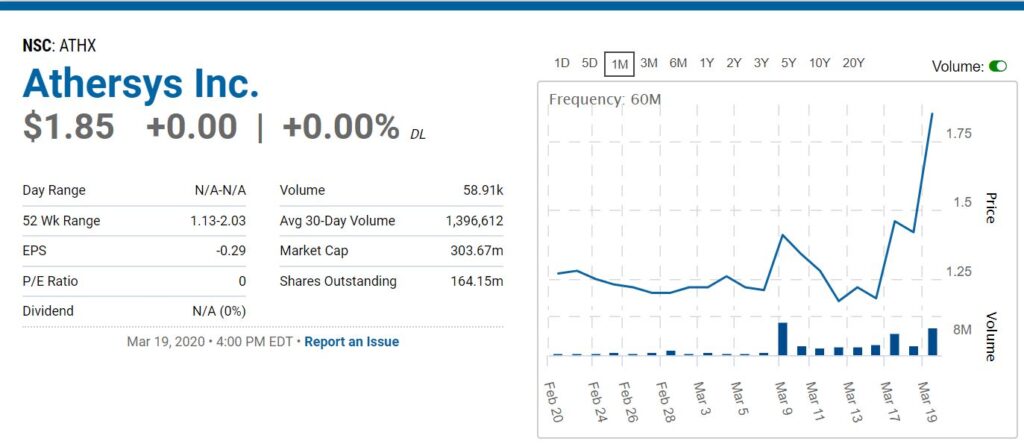
Okay, it’s a bit of a stretch, but aren’t all of these! It’s up ‘only’ 80% from a press release mentioning Corona. Maybe another press release with Corona in the headline will draw in the Corona traders. And yes, this is a hoot and a holler.
Here’s the “Whack-A-Mole” quote – this is a stem company we are familiar with from the one-year stretch we ran the Stem Cell Stock Review 3 or 4 years back.

(But first, note they ‘papered up’ Ivor Macleod, the newly-appointed Chief Financial Officer on February 3rd with 600,000 shares at $1.69 so he’s already up $276,000 – not a bad start. Inducement Grant. Good career choice Ivor.)
Headline: Athersys Announces Financial Results for Fourth Quarter and Full Year 2019
Quote #1: “The importance of this program has been reinforced by the recent COVID-19 outbreak, where many patients have subsequently become critically ill with ARDS (acute respiratory distress), which a recent World Health Organization analysis has confirmed is the primary cause of death for these patients. This often fatal syndrome can be induced by a range of pathogens, including COVID-19, SARS, MERS and virulent influenza.
UNRELATED: Our Favorite CoronaVirus Story.
Quote #2. “Other than placing patients on a ventilator, there is no effective treatment for ARDS. In January, in the early stages of the COVID-19 outbreak, as part of the U.S. Government’s COVID-19 CoronaWatch program, we were approached by BARDA in its leadership role focused on expediting diagnostics, vaccines, antivirals and therapeutic treatments for these patients. After undergoing multiple reviews, we are pleased to announce that MultiStem® was designated as a “Highly Relevant” therapeutic for COVID-19 by BARDA. We now are working to expedite the further advancement of the program, which has also shown relevance to certain other areas of interest for BARDA,” added Dr. Van Bokkelen.
Get it, got it, adding to the Watch List and we’re gone.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 that involve risks and uncertainties. These forward-looking statements relate to, among other things, the expected timetable for development of our product candidates, our growth strategy, and our future financial performance, including our operations, economic performance, financial condition, prospects, and other future events. We have attempted to identify forward-looking statements by using such words as “anticipates,” “believes,” “can,” “continue,” “could,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “should,” “suggest,” “will,” or other similar expressions. These forward-looking statements are only predictions and are largely based on our current expectations. A number of known and unknown risks, uncertainties, and other factors could affect the accuracy of these statements. Some of the more significant known risks that we face that could cause actual results to differ materially from those implied by forward-looking statements are the risks and uncertainties inherent in the process of discovering, developing, and commercializing products that are safe and effective for use as therapeutics, including the uncertainty regarding market acceptance of our product candidates and our ability to generate revenues. These risks may cause our actual results, levels of activity, performance, or achievements to differ materially from any future results, levels of activity, performance, or achievements expressed or implied by these forward-looking statements. Other important factors to consider in evaluating our forward-looking statements include: our ability to successfully finalize and implement an alliance with BARDA and the terms of any such alliance, our ability to raise capital to fund our operations; the timing and nature of results from our MultiStem clinical trials, including the MASTERS-2 Phase 3 clinical trial and Healios’ TREASURE and ONE-BRIDGE clinical trials in Japan; the possibility of delays in, adverse results of, and excessive costs of the development process; our ability to successfully initiate and complete clinical trials of our product candidates; the possibility of delays, work stoppages or interruptions in manufacturing by third parties to us, such as due to material supply constraints, contaminations, or regulatory issues, which could negatively impact our trials and the trials of our collaborators; uncertainty regarding market acceptance of our product candidates and our ability to generate revenues, including MultiStem cell therapy for the treatment of stroke, acute respiratory distress syndrome, acute myocardial infarction and trauma, and the prevention of graft-versus-host disease and other disease indications; changes in external market factors; changes in our industry’s overall performance; changes in our business strategy; our ability to protect and defend our intellectual property and related business operations, including the successful prosecution of our patent applications and enforcement of our patent rights, and operate our business in an environment of rapid technology and intellectual property development; our possible inability to realize commercially valuable discoveries in our collaborations with pharmaceutical and other biotechnology companies; our ability to meet milestones and earn royalties under our collaboration agreements, including the success of our collaboration with Healios; our collaborators’ ability to continue to fulfill their obligations under the terms of our collaboration agreements and generate sales related to our technologies; the success of our efforts to enter into new strategic partnerships and advance our programs, including, without limitation, in North America, Europe and Japan; our possible inability to execute our strategy due to changes in our industry or the economy generally; changes in productivity and reliability of suppliers; and the success of our competitors and the emergence of new competitors. You should not place undue reliance on forward-looking statements contained in this press release, and we undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise.












