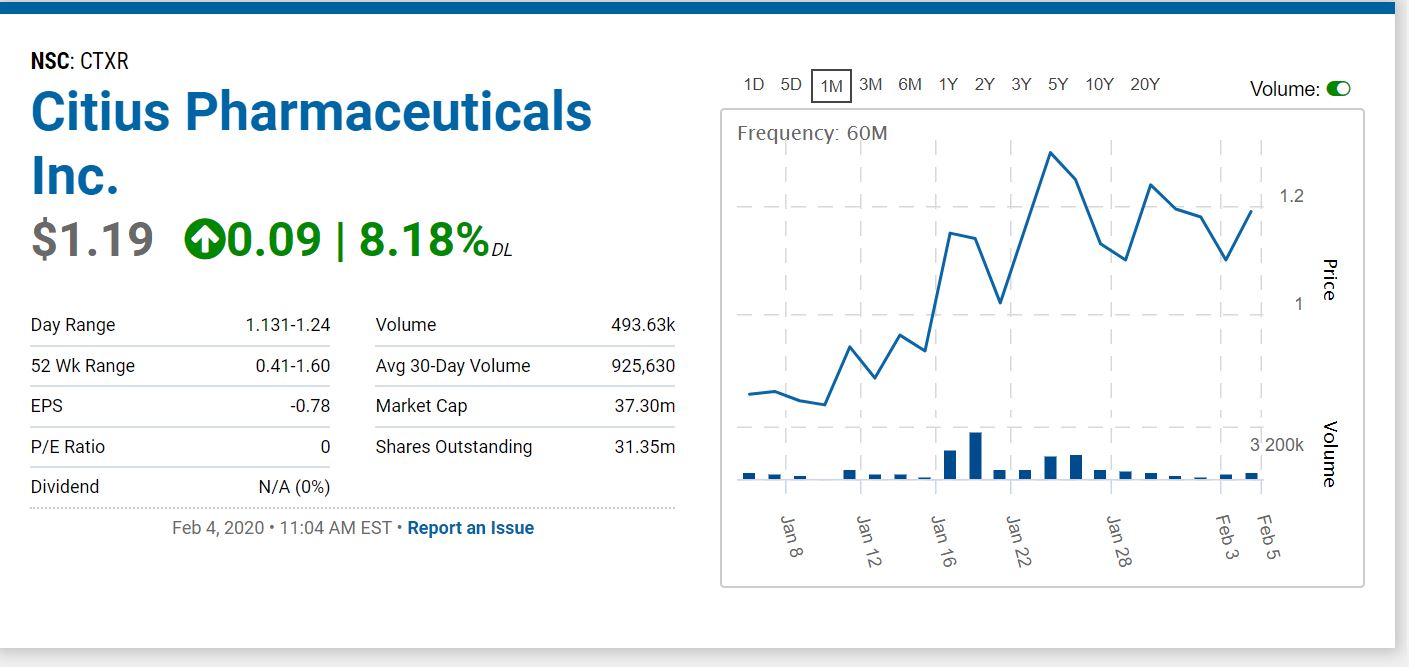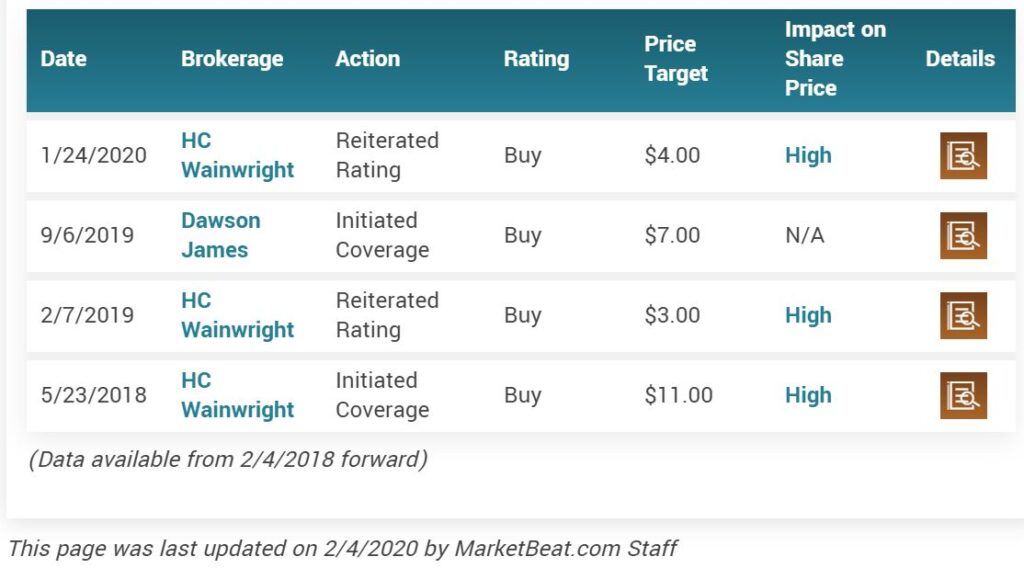
H.C. Wainwright analyst Vernon Bernardino raised the price target on Citius Pharmaceuticals (NASDAQ: CTXR) to $4.00 (from $1.65) while maintaining a Buy rating.
The analyst commented, “In September 2019, the Mino-Lok Phase 3 study Data Monitoring Committee (DMC), which is comprised of a panel of experts on bloodstream infections, conducted its first review of unblinded data after completion of enrollment of the first 40% patients in the study. One of the initial tasks of the DMC is to conduct a pre-specified interim analysis for futility in the Phase 3 study, or in other words, review study results to determine whether the primary endpoint (i.e., a significant difference between Mino-Lok therapy [MLT] versus conventional antibiotic lock therapy [ALT] in the time to catheter failure) is likely to be achieved.
We believe the recent review of the Mino-Lok Phase 3 study by its DMC, which advised Citius to proceed without any protocol changes and suggested the study remains on track to achieve its primary endpoint, was positive. We also found insights we gained regarding the catheter-related bloodstream infections (CRBSIs) market make us more optimistic regarding Mino-Lok.
Therefore, we reiterate our Buy rating with a price target of $4.00 versus our prior price target of $1.65. We think Citius shares are underappreciated ahead of a top-line Phase 3 data readout that we expect could be a positive catalyst in mid-2020.”
Biotech 5 Pack. 5 Biotech Stocks We Expect to Double in 2020.
Client, see reports for disclosure and disclaimer details.